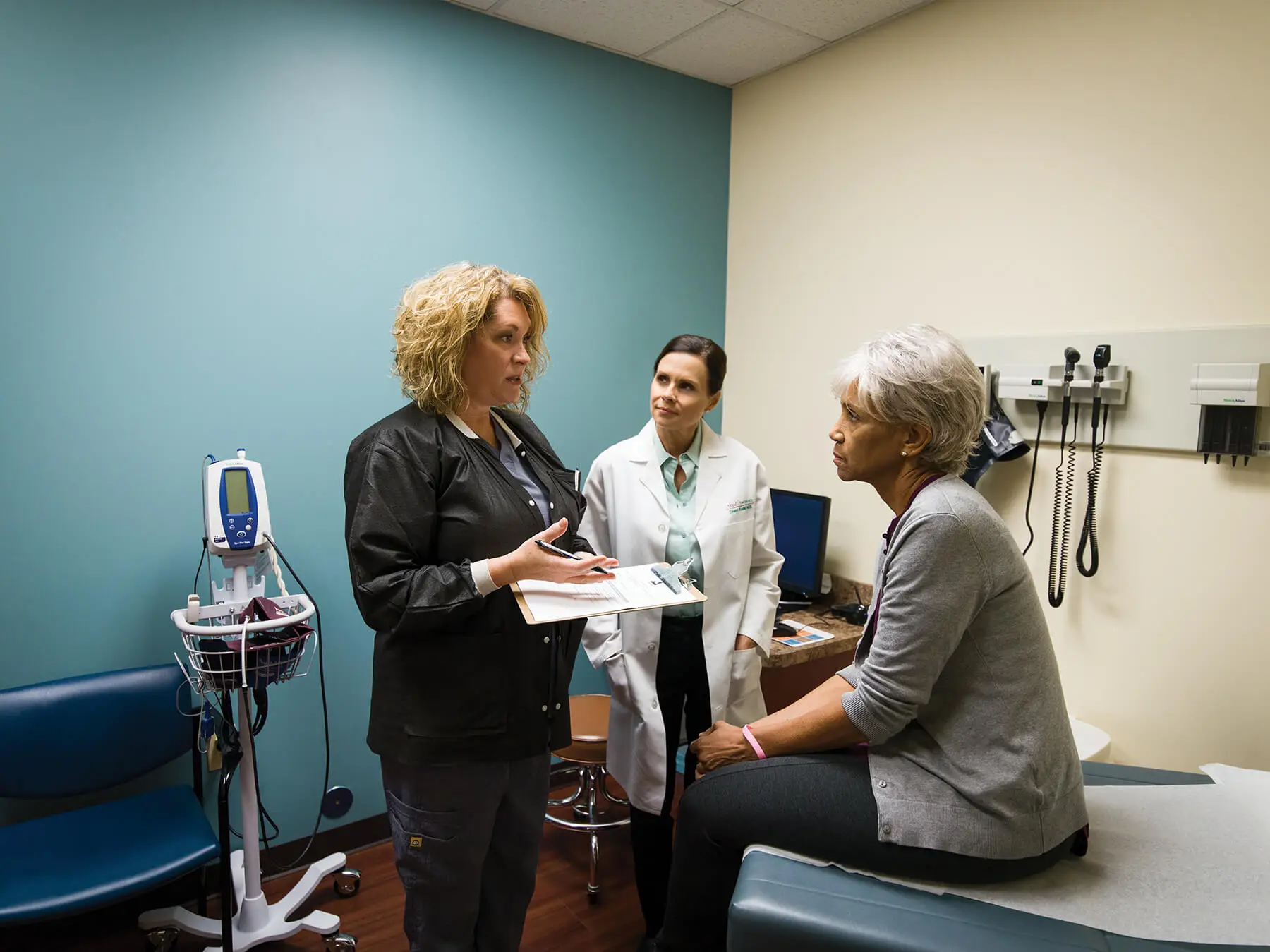
Novocure and US Oncology Research Announce Collaboration on PANOVA-3 Trial for Patients with Advanced Pancreatic Cancer
US Oncology Research will open 10 clinical trial sites, enrolling patients in Novocure’s PANOVA-3 trial to test the efficacy of Tumor Treating Fields combined with nab-paclitaxel and gemcitabine in patients with unresectable locally advanced pancreatic cancer
ST. HELIER, JERSEY, U.K. and THE WOODLANDS, TEXAS, U.S. (August 15, 2018) – Novocure (NASDAQ: NVCR) and US Oncology Research are collaborating on PANOVA-3, a phase 3 pivotal trial testing the efficacy of Tumor Treating Fields combined with nab-paclitaxel and gemcitabine in patients with unresectable locally advanced pancreatic cancer. Tumor Treating Fields is a cancer therapy that uses electric fields tuned to specific frequencies to disrupt cell division, inhibiting tumor growth and causing affected cancer cells to die. US Oncology Research, supported by The US Oncology Network – one of the largest networks of integrated, community-based oncology practices – will open 10 clinical trial sites enrolling patients in Novocure’s PANOVA-3 trial.
“Pancreatic cancer is one of the most aggressive forms of cancer and was the third leading cause of cancer death in the U.S. last year,” said Daniel D. Von Hoff, M.D., F.A.C.P., principal investigator for PANOVA-3, physician in chief, distinguished professor at the Translational Genomics Research Institute in Phoenix and the chief scientific officer for US Oncology Research. “I believe Tumor Treating Fields has the potential to improve survival in this disease. I am pleased to have US Oncology Research’s support.”
PANOVA-3 is a randomized, open-label trial that will include 556 patients with unresectable locally advanced pancreatic cancer. Patients should have an Eastern Cooperative Oncology Group (ECOG) score of 0-2 and no prior progression or treatment. Patients in the experimental arm will use a Tumor Treating Fields delivery system tuned to 150 kHz for at least 18 hours a day until the disease progresses. The primary trial endpoint is overall survival. The secondary endpoints include progression free survival, local progression free survival, objective response rate, one-year survival rate, quality of life, pain-free survival, resectability rate and toxicity.1
“Our strategic collaboration with US Oncology Research is an important milestone as we aim to bring better treatment options to patients with pancreatic cancer,” said Dr. Eilon Kirson, Novocure’s chief science officer and head of research and development. “We believe this relationship will accelerate enrollment of patients in our PANOVA-3 trial. We look forward to working with US Oncology Research and The US Oncology Network to advance cancer care.”
About Pancreatic Cancer
Pancreatic cancer was the third leading cause of cancer death in the U.S. in 2017. Five-year survival among patients with pancreatic cancer is about 8 percent. The American Cancer Society estimated that about 53,000 people would be diagnosed with pancreatic cancer and about 43,000 people would die from the disease in 2017. Worldwide, more than 330,000 people die from the disease every year.
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves approximately 60 research sites and more than 150 locations managing about 300 active trials at any given time. Physicians in the research network have enrolled more than 75,000 patients in over 1,600 trials since inception about two decades ago and have played a role in more than 75 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information, visit www.usoncology.com/oncologists.
About The US Oncology Network
Every day, The US Oncology Network (The Network) helps more than 1,400 independent physicians deliver value-based, integrated care for patients –close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About Novocure
Novocure is an oncology company developing a profoundly different cancer treatment utilizing a proprietary therapy called Tumor Treating Fields, the use of electric fields tuned to specific frequencies to disrupt solid tumor cancer cell division. Novocure’s commercialized product is approved for the treatment of adult patients with glioblastoma. Novocure has ongoing or completed clinical trials investigating Tumor Treating Fields in brain metastases, non-small cell lung cancer, pancreatic cancer, ovarian cancer and mesothelioma.
Headquartered in Jersey, Novocure has U.S. operations in Portsmouth, New Hampshire, Malvern, Pennsylvania and New York City. Additionally, the company has offices in Germany, Switzerland, Japan and Israel. For additional information about the company, please visit www.novocure.com or follow us at www.twitter.com/novocure.
Forward-Looking Statements
In addition to historical facts or statements of current condition, this press release may contain forward-looking statements. Forward-looking statements provide Novocure’s current expectations or forecasts of future events. These may include statements regarding anticipated scientific progress on its research programs, clinical trial progress, development of potential products, interpretation of clinical results, prospects for regulatory approval, manufacturing development and capabilities, market prospects for its products, coverage, collections from third-party payers and other statements regarding matters that are not historical facts. You may identify some of these forward-looking statements by the use of words in the statements such as “anticipate,” “estimate,” “expect,” “project,” “intend,” “plan,” “believe” or other words and terms of similar meaning. Novocure’s performance and financial results could differ materially from those reflected in these forward-looking statements due to general financial, economic, regulatory and political conditions as well as more specific risks and uncertainties facing Novocure such as those set forth in its Annual Report on Form 10-K filed on February 22, 2018, with the U.S. Securities and Exchange Commission. Given these risks and uncertainties, any or all of these forward-looking statements may prove to be incorrect. Therefore, you should not rely on any such factors or forward-looking statements. Furthermore, Novocure does not intend to update publicly any forward-looking statement, except as required by law. Any forward-looking statements herein speak only as of the date hereof. The Private Securities Litigation Reform Act of 1995 permits this discussion.
Media Contacts
Novocure
Ashley Cordova
212-767-7558
acordova@novocure.com
US Oncology Research
Claire Crye
281-825-9927
Claire.Crye@usoncology.com
###
More patients in suburban Chicago will benefit from the combined expertise and resources
Chicago, Illinois (August 6, 2018) – Illinois Cancer Specialists (ICS), an affiliate of The US Oncology Network (The Network), announced today that Progressive Care has joined its practice. The combined practice will continue to offer comprehensive, state-of-the-art cancer care in its nine convenient, patient-friendly community settings throughout suburban Chicago. ICS has been a part of The US Oncology Network, one of the nation’s largest networks of integrated community-based oncology practices, for several years.
“The Network will benefit greatly from the addition of these experienced oncology care providers,” said Marcus Neubauer, M.D., chief medical officer. “I am looking forward to collaborating with them as part of our larger organization in the Chicago area.”
“We’re honored to welcome the physicians and staff from Progressive Care to our practice,” said David Hakimian, M.D., ICS practice president. “This integration brings Chicagoland patients access to 22 physicians in nine of their local communities, giving them confidence that they’re getting the expertise, resources and teamwork they want and need when facing cancer.”
Clinical teams will be integrated to leverage expertise and tools that drive value-based care, and employ the latest technologies and drug therapies to ensure that patients receive high-quality care in a convenient setting. ICS treatment centers feature outpatient treatment and diagnostic facilities including chemotherapy, radiation therapy, positron emission tomography, computerized tomography, stem-cell transplant, clinical research, laboratory, x-ray studies and specialty pharmaceuticals.
“The physicians of Progressive Care have been dedicated to bringing the latest innovations to cancer care for more than 26 years,” said Mark Karides, M.D., Progressive Care practice president. “By coming together with ICS, we are able to advance cancer care even further while maintaining our focus on the compassionate, personalized patient care for which we are known throughout our community.”
By joining ICS, Progressive Care physicians can also collaborate with an expanded physician community of more than 1,400 oncologists in The US Oncology Network. As members of The Network, providers share expertise and best practices through committees, workshops, conferences and via The Oncology Portal, a secure online portal where physicians can share data and collaborate on complex cases. Progressive Care physicians will also be able to utilize Value Pathways, the evidence-based treatment guidelines developed by physicians and clinicians in The Network together with the National Comprehensive Cancer Network® (NCCN®) and other tools and technologies.
About Illinois Cancer Specialists
Illinois Cancer Specialists is dedicated to providing convenient, compassionate and leading-edge cancer care to patients in Chicago and the surrounding areas. With more than 20 physicians and nine locations, ICS brings patients advanced cancer care, along with the tools, resources and support they need to help them through their cancer journey. ICS is proud to offer more: more physician collaboration, more clinical trials and more convenient locations. Find out how patients are a priority at Illinois Cancer Specialists, by visiting www.IllinoisCancerSpecialists.com.
Illinois Cancer Specialists is an affiliate of The US Oncology Network. This collaboration unites ICS with more than 1,400 independent physicians dedicated to delivering value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The US Oncology Network is supported by McKesson Specialty Health, whose coordinated resources and infrastructure allow doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. ICS also participates in clinical trials through US Oncology Research, which has played a role in more than 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information, visit www.usoncology.com.
Media Contact:
Christy Sullivan
Illinois Cancer Specialists
(847) 421-4919
Christy.Sullivan@usoncology.com
###
Medical oncologist is appointed to spearhead The Network’s significant medical programs and collaborative, integrated approach to cancer care
The Woodlands, Texas (July 17, 2018) —The US Oncology Network (The Network) announced the appointment of Marcus Neubauer, M.D., as its chief medical officer, building on its mission of delivering a comprehensive approach to cancer care. Michael Seiden, M.D., Ph.D. will continue to lead The Network as its president.
Dr. Neubauer has been a practicing medical oncologist for over 20 years, serving previously as a physician and leader in our Kansas City practice. For the past five years he has served as the vice president and medical director for payer, clinical services, and value-based care, working with over 1,400 community oncologists across the country. He is a recognized leader in innovation in clinical pathways, value-based care and contracting and the Center for Medicare and Medicaid Innovation’s (CMMI®) Oncology Care Model.
Dr. Neubauer has served numerous appointments and committees, received honors, awards and has been featured as a thought leader in publications for the past 25 years. While serving The US Oncology Network, he has continued his clinical activities in Seattle as an associate clinical professor for the Seattle Cancer Care Alliance.
“I am thrilled to be continuing my career with The US Oncology Network in this leadership role,” said Dr. Neubauer. “It’s an exciting opportunity for me to help guide The Network’s approach to cancer treatment amid the many changes in the healthcare environment to ensure comprehensive high-quality, value-based care in the community setting.”
Dr. Neubauer has been the lead or contributing author on more than 60 original, peer-reviewed research articles, editorials and meeting abstracts. In addition to his clinical work and medical-director role for The Network, he has served on the Journal of Oncology Practice editorial board for six years and is a current editorial board member for the Community Oncology and Supportive Care Journal since 2012.
“After a national search for a chief medical officer, we are excited to share that our well-respected physician-leader will serve in this forward-thinking role,” said Dr. Michael Seiden. “Dr. Neubauer will work closely with Dr. Lucy Langer, chair of The US Oncology Network national policy board and joins my executive committee effective July 16.”
Dr. Neubauer is a board-certified, fellowship-trained medical oncologist who earned his medical degree and completed his internship and residency at the University of Kansas in Kansas City. He completed his oncology fellowship at the University of Washington in Seattle. Dr. Neubauer will establish residence and office in The Woodlands, TX.
About The US Oncology Network
The US Oncology Network is one of the nation’s largest networks of integrated, community-based oncology practices dedicated to advancing high-quality, evidence-based cancer care. A physician-led organization, The US Oncology Network unites like-minded physicians and clinicians around a common vision of improving patient outcomes and quality of life. Leveraging healthcare information technology, shared best practices, evidence-based guidelines and quality measurements, physicians within The US Oncology Network are pioneering new ways to achieve this vision. The US Oncology Network is committed to strengthening patient access to integrated care in local communities across the nation, including collaboration with a variety of payers, hospitals and academic institutions. The US Oncology Network is supported by McKesson Specialty Health, a division of McKesson Corporation focused on empowering a vibrant and sustainable community patient care delivery system. For more information, visit www.usoncology.com.
About McKesson Specialty Health
McKesson Specialty Health, a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, biopharma and payer solutions, McKesson Specialty Health focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit www.mckessonspecialtyhealth.com.
Media Contact
Edie DeVine
GCI Health
209-814-9564
Edie.DeVine@gcihealth.com
###
Renowned breast surgeon appointed to spearhead The Network’s surgical programs and collaborative, integrated approach to cancer care
The Woodlands, Texas (July 9, 2018) —The US Oncology Network (The Network) announced the appointment of Loren Rourke, M.D., M.H.C.M., as its chief surgical officer, a new role created to support and enhance the surgical community within The Network, fortifying its mission of delivering comprehensive approach to cancer care.
Dr. Rourke brings more than 15 years of surgical experience to The US Oncology Network – experience that includes numerous academic appointments, committees, honors, awards and publications. Most recently, she served as regional breast program director at Houston Methodist. Previously, she held such leadership positions as medical director for the Maine Medical Center Cancer Institute Breast Center (Scarborough, ME) and surgical director of St. Luke’s Woodlands breast program.
“I am thrilled to be joining The US Oncology Network in this groundbreaking, new role,” said Dr. Rourke. “It’s an exciting opportunity for me to help guide The Network’s approach to cancer treatment – an approach that is team-based, comprehensive and recognizes the pivotal role surgeons play in treating cancer.”
Dr. Rourke has contributed to the development of new surgical techniques in breast surgical oncology, as the author or co-author of more than ten original, peer-reviewed research articles, editorials and book chapters. In addition to her clinical work, she has lectured nationally and internationally and is the recipient of several scholarships and awards.
With more than 60 affiliated practices and 1,400 physicians, The US Oncology Network’s affiliated practices treated almost one million patients in the year ending March 31, 2018. Surgeons have played an important role in The Network’s approach to integrated cancer care, an area that continues to grow in its importance.
“We are proud to welcome Dr. Rourke to The US Oncology Network in a unique and forward-thinking role,” said Michael Seiden, president and chief medical officer. “Bringing in a surgeon of her caliber emphasizes The Network’s strategic direction towards truly comprehensive care that includes surgery, medical oncology and radiation oncology. With more than 300 surgeons in The Network, Dr. Rourke will be a key leader working closely with me and the entire US Oncology leadership team.”
Dr. Rourke is a board-certified fellowship-trained breast surgeon who earned her medical degree from Louisiana State University School of Medicine in Shreveport, LA. She completed her internship at Indiana State University School of Medicine in Indianapolis, IN, surgery residency at University of Tennessee Graduate School of Medicine in Knoxville, TN, and later completed her interdisciplinary, breast clinical fellowship at The University of Texas MD Anderson Cancer Center in Houston, TX. Dr. Rourke will be based at the headquarters for McKesson Specialty Health in The Woodlands, TX.
About The US Oncology Network
The US Oncology Network is one of the nation’s largest networks of integrated, community-based oncology practices dedicated to advancing high-quality, evidence-based cancer care. A physician-led organization, The US Oncology Network unites like-minded physicians and clinicians around a common vision of improving patient outcomes and quality of life. Leveraging healthcare information technology, shared best practices, evidence-based guidelines and quality measurements, physicians within The US Oncology Network are pioneering new ways to achieve this vision. The US Oncology Network is committed to strengthening patient access to integrated care in local communities across the nation, including collaboration with a variety of payers, hospitals and academic institutions. The US Oncology Network is supported by McKesson Specialty Health, a division of McKesson Corporation focused on empowering a vibrant and sustainable community patient care delivery system. For more information, visit www.usoncology.com.
About McKesson Specialty Health
McKesson Specialty Health, a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, biopharma and payer solutions, McKesson Specialty Health focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit www.mckessonspecialtyhealth.com.
Media Contact
Edie DeVine
GCI Health
209-814-9564
Edie.DeVine@gcihealth.com
###
Expansive clinical research and real-world evidence data to be presented on prostate cancer, genome sequencing, melanoma, bladder cancer, metastatic breast cancer and more
The Woodlands, Texas (May 30, 2018) – McKesson Specialty Health announced today that oncologists from The US Oncology Network (The Network) and US Oncology Research will showcase detailed findings from 43 studies during the 54th Annual Meeting of the American Society of Clinical Oncology (ASCO), held June 1–5, 2018, in Chicago with more than 32,000 oncology professionals in attendance. The study abstracts accepted for presentation represent substantial contributions made by community oncologists towards the understanding and advancement of cancer care.
“Research conducted by community oncologists and presented at this year’s ASCO meeting demonstrates the dedication of these professionals in not only advancing investigational treatment options, but also offering cutting-edge research to their patients,” said Michael Seiden, M.D., Ph.D., president and chief medical officer for The US Oncology Network and US Oncology Research. “There have been tremendous advancements in cancer care, and being selected for a clinical study is no longer seen as a last option, but rather as access to some of the latest investigational therapies for treating a wide range of cancers. Our community-based oncology network, made up of 1,400 independent physicians, allows access to resources and emerging treatments across the country. This meeting provides a time to focus on the unmet needs of patients through clinical trials that are leading the way in the fight against cancer.”
Key study presentations will include topics such as genome sequencing in lung cancer, metastatic breast cancer, real-world evidence in bladder cancer and melanoma, and Phase 3 data from the PROTECT study in asymptomatic or minimally symptomatic metastatic, castration-resistant prostate cancer.
“The ability for community oncologists to participate in groundbreaking cancer research must remain a high priority for all research organizations, including those programs supported by the National Cancer Institute,” said Nicholas J. Vogelzang, M.D., FASCO, FACP, medical oncologist with Comprehensive Cancer Centers of Nevada, an affiliate of The US Oncology Network, and 2018 recipient of an OncLive Giants of Cancer Care award. “It is critically important for oncologists around the world, regardless of the type of center they are practicing in, to be able to find time to conduct research and make advancements in cancer care. I’m proud to be an active clinical investigator within The US Oncology Network, where several studies have been selected for presentation at this year’s ASCO annual meeting. This conference is a time for us to share knowledge and gain insights to better and more effectively treat patients, while contributing to our collective mission to eradicate cancer.”
Key oral presentations will include:
- Results of PROTECT: A randomized phase 3 trial of PROSTVAC-V/F (PRO) in men with asymptomatic or minimally symptomatic metastatic, castration-resistant prostate cancer
Abstract #: 5006
Date/Time: Monday, June 4, 3-6:00 p.m.
Location: Hall D1
Affiliated Author: Nicholas J. Vogelzang, M.D., FASCO, FACP, Comprehensive Cancer Centers of Nevada, US Oncology Research
- Genome-wide sequencing for early stage lung cancer detection from plasma cell-free DNA (cfDNA): The Circulating Cancer Genome Atlas (CCGA) studyAbstract #: LBA8501
Date/Time: Monday, June 4, 8-11:00 a.m.
Location: Hall B1
Affiliated Author: Donald Richards, M.D., Ph.D., Texas Oncology, The US Oncology Network
- Efficacy of sacituzumab govitecan (anti-Trop-2-SN-38 antibody-drug conjugate) for treatment-refractory hormone-receptor positive (HR+)/HER2- metastatic breast cancer (mBC)
Abstract #: 1004
Date/Time: Sunday, June 3, 8-11:00 a.m.
Location: Hall D2
Affiliated Author: Joyce O’Shaughnessy, M.D., Texas Oncology, The US Oncology Network, US Oncology Research
- First results from the primary analysis population of the phase 2 study of erdafitinib (ERDA; JNJ-42756493) in patients (pts) with metastatic or unresectable urothelial carcinoma (mUC) and FGFR alterations (FGFRalt)
Abstract #: 4503
Date/Time: Sunday, June 3, 8–-11:00 a.m.
Location: Arie Crown Theater
Affiliated Author: Mark T. Fleming, M.D., Virginia Oncology Associates, US Oncology Research
- Prevalence of clonal hematopoiesis of indeterminate potential (CHIP) measured by an ultra-sensitive sequencing assay: Exploratory analysis of the Circulating Cancer Genome Atlas (CCGA) study
Abstract #: 12003
Date/Time: Tuesday, June 5, 8–11:00 a.m.
Location: S406
Affiliated Author: Michael Seiden, M.D., Ph.D., The US Oncology Network, US Oncology Research
An additional 29 poster presentations and nine published abstracts affiliated with US Oncology Research will be featured as part of the ASCO program. The full schedule of US Oncology Research–affiliated data presentations, including location information, can be found here.
For more information or to be put in touch with a trial investigator, please contact Edie DeVine at 209-814-9564 or Edie.DeVine@gcihealth.com. Please visit Booth #5123 on the Main Floor at ASCO.
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves approximately 60 research sites and nearly 160 locations managing about 300 active trials at any given time. Physicians in the research network have enrolled more than 73,000 patients in more than 1,600 trials since inception in 1992 and have played a role in more than 70 FDA-approved cancer therapies, about one-third of all cancer therapies approved by the FDA to date. For more information call 800-482-6700 or visit www.usoncology.com/oncologists.
About The US Oncology Network
The US Oncology Network is one of the nation’s largest networks of integrated, community-based oncology practices dedicated to advancing high-quality, evidence-based cancer care. A physician-led organization, The US Oncology Network unites like-minded physicians and clinicians around a common vision of improving patient outcomes and quality of life. Leveraging healthcare information technology, shared best practices, evidence-based guidelines and quality measurements, physicians within The US Oncology Network are pioneering new ways to achieve this vision. The US Oncology Network is committed to strengthening patient access to integrated care in local communities across the nation, including collaboration with a variety of payers, hospitals and academic institutions. The US Oncology Network is supported by McKesson Specialty Health, a division of McKesson Corporation focused on empowering a vibrant and sustainable community patient care delivery system. For more information, visit www.usoncology.com.
About McKesson Specialty Health
McKesson Specialty Health, a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, biopharma and payer solutions, McKesson Specialty Health focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit www.mckessonspecialtyhealth.com.
Media Contact
Edie DeVine
GCI Health
209-814-9564
Edie.DeVine@gcihealth.com
###
Study Wins the 2018 Clinical and Research Excellence (CARE) Award for Best in Health Economics and Outcomes
The Woodlands, Texas (May 16, 2018) — A powerful, real-world evidence study designed by McKesson Specialty Health for its biopharma partner played a key role in the Food and Drug Administration’s (FDA) approval for first-line indication for a rare disease. Recently “Merkel Cell Carcinoma Observational Study for Historic Comparison” received the 2018 Clinical and Research Excellence (CARE) Award for Best in Health Economics and Outcomes Research, recognizing how data research supports true innovation in oncology care.
The award acknowledges excellence in health economics and outcomes research (HEOR), a strategic focus for the company, and was announced at an award ceremony with organizers Pharma Intelligence, biopharma companies, researchers, and providers last month in Boston.
Clinical trial data has historically been the way to earn first-line approvals of drugs by the FDA. With the growth of big data and advanced systems like electronic health records (EHRs), McKesson strategically supports advancing specialty pharmaceutical development. The success of this study highlights how real-world evidence from EHRs empowers biopharma companies to innovate, bringing new therapies and treatment options to providers and patients. The study leveraged iKnowMedSM, McKesson’s award-winning EHR that combines analytics expertise and physicians’ knowledge for accelerating cancer treatments. Numerous HEOR studies are currently being conducted by McKesson and are expected to lead to other oncology breakthroughs.
“Real-world evidence is increasingly being leveraged to support FDA approvals, enabling more therapy options to advance and optimize cancer care,” said Nicholas Robert, M.D., Medical Director, Health Economic and Outcomes Research/Health Informatics, McKesson Specialty Health and The US Oncology Network. “This award underscores the important role real-world evidence plays in improving patient outcomes by providing supporting data to clinical trial results.”
In February 2018, iKnowMed Generation 2 was recognized by Black Book Research as the top EHR for oncology and hematology for patient health data management and administrative processing, interoperability, communications, connectivity, results review/management, and decision support. It achieved the top score in 10 of the 18 EHR performance categories.
For more detailed information on how real-world evidence led to a first-line FDA approval for the Metastatic Merkel Cell Carcinoma Drug read the white paper.
About McKesson Specialty Health
McKesson Specialty Health (“MSH”), a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, manufacturer and payer solutions, MSH focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit http://www.mckessonspecialtyhealth.com/.
About The US Oncology Network
Every day, The US Oncology Network (“The Network”) helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit http://www.usoncology.com/.
# # #
Media Contact
Edie DeVine, GCI Health on behalf of McKesson Specialty Health
209-814-9564
edie.devine@gcihealth.com
Industry Recognition Highlights Texas Oncology’s Integrated Pharmacy Approach to Attain Better Patient Outcomes and Lower Costs
DALLAS (May 7, 2018) — Texas Oncology, a practice in The US Oncology Network (The Network), received a 2018 Excellence Award from Pharmacy Benefit Management Institute® (PBMI) for its Medically Integrated Pharmacy Management (MIPM) approach to achieve better results in cancer care. The MIPM model includes a team of board-certified oncologists, as well as other healthcare professionals, to deliver patient-focused solutions that drive industry-leading outcomes and contain costs.
Oral chemotherapy is increasingly important in cancer care with more than 70 FDA-approved oral chemotherapy cancer drugs. Additionally, an estimated 35 percent of new drugs by pharmaceutical manufacturers are oral drugs. Adherence to patient oral medications, therefore, is vital to successful cancer care. The national average for adherence to oncology medications is below 70 percent, but the weighted average is 96 percent for patients managed by Texas Oncology’s medically integrated pharmacy team. Furthermore, the Congressional Budget Office estimates that for every five-percentage point increase in adherence (measured by number of prescriptions), total medical costs decline by one percent.
“A medically integrated care approach with physicians, nurses, pharmacists, nutritionists, and financial counselors working together improves patient experiences and compliance while reducing costs from less waste, fewer side effects, and fewer hospitalizations,” said Jim Schwartz, executive director, pharmacy operations, Texas Oncology Pharmacy. “We are grateful for this award and for our amazing team members who provide active, patient-centered care throughout the treatment process.”
“The PBMI award reflects years of work from our dedicated Texas Oncology team,” said Steve Paulson, M.D., president and chairman of the Board, Texas Oncology. “With the help and shared knowledge gained from our peers within Texas Oncology and support from The US Oncology Network, we are improving patient adherence and lowering medical costs – particularly in oral chemotherapy.”
“The PBMI Excellence award is outstanding recognition of Texas Oncology’s leadership in advancing the Medically Integrated Pharmacy Management model,” said John Priskorn, vice president of self-administered products and services for McKesson Specialty Health, which supports The Network. “Texas Oncology’s results demonstrate that this patient-centric model is highly effective and represents a compelling vision for the future of community-based cancer care. We are proud of the Texas Oncology team, and it is a privilege to provide operational and clinical support to their medically integrated pharmacies.”
About Texas Oncology
As an independent oncology practice with more than 420 physicians and 175 locations, Texas Oncology’s community-based care offers patients high-quality cancer care without compromise, including access to leading-edge technology, advanced treatment options, and access to clinical trials. As a primary participant in US Oncology Research, Texas Oncology played a role in more than 70 FDA-approved therapies, about one-third of all cancer therapies approved by the agency to date.
Texas Center for Proton Therapy, Texas Breast Specialists, and Texas Center for Interventional Surgery are part of Texas Oncology. Texas Oncology is a member of The US Oncology Network, one of the nation’s largest community-based cancer treatment and research networks. For more information, visit www.TexasOncology.com.
About The US Oncology Network
Every day, The US Oncology Network (The Network) helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit http://www.usoncology.com/.
Media Contact
Stephanie Wick, Texas Oncology
214-665-1329
###
Renowned breast surgeon joins practice with more than two decades of experience, expanding the service offering to more patients
Fairfax, Va. (March 12, 2018) —Virginia Cancer Specialists (VCS) announced that Hernan Vargas, MD, has joined the practice. VCS, a practice in The US Oncology Network, is a premier cancer care and research center in Northern Virginia with more than 40 years of service to patients.
Dr. Vargas joins VCS with more than 20 years of experience in the care of women with breast cancer and benign conditions of the breast. Previously, he served as Associate Professor of Surgery at UCLA as well as the Director of the Breast Programs at Harbor-UCLA in Los Angeles and at Inova Fair Oaks Hospital in Fairfax, Virginia. He has contributed to the development of new surgical techniques in breast cancer care, being the author of more than 100 scientific publications. Most importantly, Dr. Vargas’ goal is to provide high-quality, patient-centered care within a multidisciplinary group of like-minded individuals.
“Breast cancer patients meet us during one of the most vulnerable times in their lives,” said Dr. Vargas. “For those of us who see this as a calling and not just a job, we have the privilege of helping patients, guiding them at this time when they need it most.”
In addition to his clinical work, Dr. Vargas has lectured nationally and internationally. He is the recipient of several awards for leadership and teaching excellence and has been recognized as a “Top Doctor” in breast surgery by Washingtonian Magazine, LA Magazine and Castle-Connolly’s America’s Top Doctors for Cancer.
“We are proud to welcome Dr. Vargas to our exceptional breast care team,” said Linda Anderson, executive director, VCS. “His expertise and patient-centered focus will be extraordinary additions to our new line of breast surgery services offered to patients as part of our 360-degree approach to patient care.”
Dr. Vargas is a board-certified surgeon who attended the Alberto Hurtado School of Medicine in Lima, Peru. He completed his residency at UCLA Medical Center in Los Angeles as well as his fellowship at The National Cancer Institute of Health in Bethesda, Maryland. He is currently treating patients at the VCS Fair Oaks location. Call 703-208-9350 for an appointment.
About Virginia Cancer Specialists
For more than 40 years, Virginia Cancer Specialists (VCS) has contributed to the campaign against cancer and diseases of the blood. VCS has nine locations throughout Northern Virginia staffed by highly-skilled physicians, each delivering exceptional care. The practice has built a world-class treatment team of cancer specialists, as well as acquiring the very latest treatment technology to help achieve the best outcomes for patients. VCS unites medical and radiation oncologists, breast and musculoskeletal surgeons, hematologists, palliative care physicians, oncology nurse navigators, oncology infusion nurses, radiation therapists, genetic counselors, oncology pharmacists and laboratory technicians. All working together, this team designs the optimal multidisciplinary treatment program, efficiently shares knowledge, executes the treatment plan, and provides a patient-centered, consumer-friendly approach to cancer care. Patients receive the full spectrum of high-quality care necessary to treat their disease from a care team united in their effort to provide each patient with the specific personalized care they need to battle cancer. The physicians at VCS have published more than 100 abstracts and papers over the last few years and have been invited to present at national meetings. To learn more about Virginia Cancer Specialists, please visit www.VirginiaCancerSpecialists.com.
Virginia Cancer Specialists is an affiliate of The US Oncology Network (The Network). This collaboration unites VCS with more than 1,400 independent physicians dedicated to delivering value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The US Oncology Network is supported by McKesson Specialty Health, whose coordinated resources and infrastructure allow doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. VCS also participates in clinical trials through US Oncology Research, which has played a role in more than 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information, visit www.usoncology.com.
Media Contacts
Michelle Keene, Virginia Cancer Specialists
571-499-1914
Michelle.Keene@usoncology.com
Claire Crye, The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
###
Practice increases clinical trial and advanced treatment options while gaining access to resources that improve operational efficiencies
Philadelphia, Pa. (January 18, 2018) – Consultants in Medical Oncology and Hematology (CMOH), a premier provider of comprehensive medical oncology services throughout the Philadelphia region for nearly 30 years and a national leader in the development of patient and physician-focused value-based cancer care delivery models, has joined The US Oncology Network (The Network). The Network is the largest organization of its kind comprised of community-based oncology practices dedicated to advancing high-quality, evidence-based cancer care.
By joining The Network, CMOH gains access to some of the most advanced oncology treatments available today, as well as innovative, proven solutions that can strengthen the practice’s ability to deliver high-quality, value-based care in convenient community locations. With the addition of CMOH, The Network extends its presence in the Northeast, while also enhancing its expertise in alternative, patient-focused, value-based payment models.The Network is supported by McKesson Specialty Health, an industry leader in solutions that empower the community patient care delivery system to advance the science, technology and quality of care.
“The Philadelphia market has a reputation for strong community-based oncology,” said Kirk Kaminsky, president, The US Oncology Network and Provider Services, McKesson Specialty Health. “We’re excited to bring The Network’s model to CMOH in addition to the other support services we provide Philadelphia-based oncologists.”
“Uniting with The US Oncology Network will further fuel our goal of the high-quality, patient-centered care that we have always provided, and enable us to offer even more advanced treatment options not usually available in a community setting,” said John D. Sprandio, M.D., medical oncologist and chief of Medical Oncology and Hematology at CMOH. “The vast resources of The Network will be invaluable in helping us continue to advance our agenda of providing progressively better care and quality of life for our patients while reducing unnecessary spending through improved care delivery. This is increasingly important to employers, local and national payers, as well as patients and their families who are facing increasing costs and rising out of pocket expenses in pursuit of the highest quality of cancer care.”
By joining The Network, CMOH can collaborate with their colleagues across America through eTumor Boards and The Oncology Portal, drawing from the collective knowledge and expertise of more than 1,400 physicians in The Network. Additionally, by uniting with The Network, the practice gains the ability to offer patients novel clinical trials through US Oncology Research, a leader in bringing innovative therapies to patients in local communities across the nation. The organization has played a role in approximately 70 FDA-approved cancer therapies, about one-third of all cancer therapies approved by the FDA to date.
“Dr. Sprandio and his colleagues are changing the way cancer care is delivered with their innovative care delivery model, the Oncology Patient Centered Medical Home®, that improves patient care, provides a better patient experience and lowers costs through more effective care delivery,” said Chad Warnick, regional senior vice president at McKesson Specialty Health. “We are excited to have the opportunity to collaborate with and learn from them, as they share our common goal of advancing patient-focused cancer care while keeping community practices strong and vibrant in today’s value-based environment.”
With nine physicians and one advanced practice provider, CMOH offers patients and their families an enhanced level of services and cutting-edge protocols. The practice has four state-of-the-art treatment centers, with offices in Broomall, Glen Mills, Newtown Square and Exton. CMOH provides highly personalized, coordinated care based on a unique nationally recognized care delivery model designed by Dr. Sprandio, a recognized leader in the development of patient-centric, value-based care models. The practice developed propriety software (Iris™ Clinician Suite) and standardized processes to allow for its highly efficient, personalized approach to care, stressing patient engagement and communication to keep patients active and fully informed in every step of their treatment journey. The Iris™ Clinician Suite is central to the practice’s success in relentlessly driving the consistency, quality and value of cancer care.
Care is delivered by a fully accredited team of medical oncologists, nurses, patient navigators, and support staff. These highly-skilled compassionate professionals not only provide medical oncology services, but also coordinate and manage all of the patient’s tests, external appointments and communication with other doctors, handling these burdensome tasks which can be challenging to patients.
About Consultants in Medical Oncology and Hematology
Since Consultants in Medical Oncology and Hematology (CMOH) was founded almost 30 years ago, it has become a leading oncology and hematology practice. Care delivery is based on the nationally recognized model developed by practice leader, Dr. John Sprandio, stressing personalized, high-quality coordinated cancer treatment. The practice provides patients access to the most advanced and innovative protocols and treatments available today, operating from four locations throughout the greater Philadelphia area. CMOH has over 75 staff members, including oncology nurse practitioners, physician assistants, protocol nurse managers, certified chemotherapy nurses, patient navigators and patient financial consultants. At the core of the practice’s success is a single-minded focus on providing a high-level of service to their patients and families as well as referring and co-managing physicians. The services provided or coordinated by CMOH are highly recognized in the Philadelphia market, based on patient satisfaction surveys, National Cancer Center Network guideline audits, National Committee for Quality Assurance recognition, their performance in the (CMS) Oncology Care Model, and internal audits.
About The US Oncology Network
Every day, The US Oncology Network (The Network) helps more than 1,400 independent physicians deliver value-based, integrated care for patients—close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
Media Contact
Claire Crye
The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
###
Providers using iKnowMed certified EHR technology are eligible for Quality Performance bonus points under Merit-based Incentive Payment System
The Woodlands, Texas (January 5, 2018) — McKesson Specialty Health, an industry leader in services and technologies that advance community-based healthcare, announced that iKnowMedSM electronic health record (EHR) (version 6.8) and iKnowMed Generation 2 EHR (version 3.0) have received ONC-ACB 2015 Edition Certification. These products, with the support of My Care PlusSM patient portal (version 3.13) and Practice InsightsSManalytics (version 1.5), were certified as “Meaningful Use-ready” to support practices participating in value-based care. In addition to the many clinical and operational benefits these technologies bring to practices, providers using iKnowMed are eligible for bonus points in the Quality Performance category under the Merit-based Incentive Payment System (MIPS).
The products were tested and certified through the Drummond Group’s Electronic Health Records Office of the National Coordinator Authorized Certification Body (ONC-ACB) program and are now certified as 2015 Edition compliant. Practices utilizing iKnowMed can demonstrate Meaningful Use in 2015, helping them avoid penalties under the American Recovery and Reinvestment Act while making them eligible for incentive payments under MIPS.
Combining the advanced technologies of McKesson Specialty Health with expertise from physicians in The US Oncology Network, these leading-edge technology products provide unique support for practices, enabling them to efficiently respond to the ever-changing requirements of the value-based environment while delivering high-quality care to patients. The products incorporate the latest interoperability standards, facilitating seamless exchange of patient information with other electronic systems, including EHRs at collaborating practices and hospitals.
“Our 2015 Edition certified products are leading the way in value-based care solutions, providing efficient technologies that drive better care and care coordination, support improved clinical and business decisions, and enhance patient engagement,” said Dan Lodder, vice president and general manager, Technology Solutions, McKesson Specialty Health. “Additionally, practices using iKnowMed are eligible to collect incentive dollars by attesting to Meaningful Use in 2015 and beyond, contributing to the financial success of the practice.”
The iKnowMed EHR team is committed to providing superior service and support for community-based providers, enabling practices to quickly and efficiently implement new technologies to meet Meaningful Use requirements while maximizing incentives available through MIPS. In 2015 and 2016, more than 90% of practices using iKnowMed in both The US Oncology Network and McKesson’s Onmark Select Program were successful in attesting to Meaningful Use. This provided $6.9 million in iKnowMed incentive payments to affiliated practices in 2015 and 2016.
“Maintaining compliance in today’s dynamic regulatory landscape is increasingly challenging for community-based oncology practices,” said Marcus Neubauer, M.D., vice president and medical director, Payer and Clinical Services, McKesson Specialty Health. “McKesson’s Meaningful Use certified technologies play a vital role in enabling practices to keep up to date with the changing healthcare landscape, allowing them to efficiently and successfully implement the changes that are vital to succeed in value-based care.”
About McKesson Specialty Health
McKesson Specialty Health, a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, biopharma and payer solutions, McKesson Specialty Health focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit http://www.mckessonspecialtyhealth.com/.
About The US Oncology Network
Every day, The US Oncology Network helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@mckesson.com
# # #
19 abstracts on clinical research conducted by top affiliated investigators are scheduled for presentation
The Woodlands, Texas (December 4, 2017) – At the 59th American Society of Hematology Annual Meeting and Exposition (ASH), leading investigators from The US Oncology Network and US Oncology Research will showcase detailed findings from 19 studies (four orals, 14 poster presentations and one e-publication) covering topics such as multiple myeloma, chronic lymphocytic leukemia, myeloproliferative neoplasms and more. The 59th ASH Annual Meeting, a premier scientific event in malignant and non-malignant hematology, will be held December 9-12 in Atlanta.
“The research and breakthroughs in oncology and hematology coming from community oncologists are significantly advancing treatment options for cancer,” said Michael Seiden, MD, PhD, chief medical officer for The US Oncology Network and US Oncology Research. “The commitment to current and future patients from these physicians is astounding. We are proud to take part in this important meeting, and I’m looking forward to this year’s presentations at ASH.”
Habte Yimer, MD, medical oncologist and hematologist with Texas Oncology, a practice in The US Oncology Network, co-authored a study being presented on chronic lymphocytic leukemia titled, “Improvements in Health-Related Quality of Life and Symptoms in Patients with Previously Untreated Chronic Lymphocytic Leukemia: Results from the Phase II GIBB Study of the Combination of Obinutuzumab and Bendamustine.” The oral presentation will take place Monday, December 11 at 11:30am ET in Bldg. C, Lvl. 2, C208-C210 (Georgia World Congress Center).
“In previous reporting of the results in June of 2017, we have shown the Obinutuzumab/Bendamustine combination to be safe and has significant activity in first-line setting,” said Dr. Yimer. “We will see if those results translate into clinically meaningful improvement in health-related quality of life in this year’s ASH presentation.”
Additionally, Dr. Yimer will present “Results of an Interim Safety Analysis of a Phase 2 Study of Daratumumab (Dara) Plus Cyclophosphamide, Bortezomib, and Dexamethasone (CyBorD) in Previously Untreated and Relapsed Patients (Pts) with Multiple Myeloma (MM).” The poster abstract will be available Saturday, December 9 from 5:30-7:30pm ET in Bldg. A, Lvl. 1, Hall A2.
The full schedule of US Oncology Research affiliated data presentations, including location and author information, can be found here. For more information or to interview a trial investigator, contact Claire Crye at 281-825-9927 or Claire.Crye@usoncology.com or Kerry Sinclair at 323-602-1096 or Kerry.Sinclair@gcihealth.com.
About US Oncology Research
US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves approximately 60 research sites and more than 165 locations managing about 300 active trials at any given time. Physicians in the research network have enrolled more than 70,000 patients in over 1,500 trials since inception about two decades ago and have played a role in more than 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information visit www.usoncology.com/oncologists.
About The US Oncology Network
Every day, The US Oncology Network helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About McKesson
McKesson Corporation, currently ranked 6th on the FORTUNE 500, is a global leader in healthcare supply chain management solutions, retail pharmacy, community oncology and specialty care, and healthcare information technology. McKesson partners with pharmaceutical manufacturers, providers, pharmacies, governments and other organizations in healthcare to help provide the right medicines, medical products and healthcare services to the right patients at the right time, safely and cost-effectively. United by our ICARE shared principles, our employees work every day to innovate and deliver opportunities that make our customers and partners more successful — all for the better health of patients. McKesson has been named the “Most Admired Company” in the healthcare wholesaler category by FORTUNE, a “Best Place to Work” by the Human Rights Campaign Foundation, and a top military-friendly company by Military Friendly. For more information, visit www.mckesson.com.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@usoncology.com
###
Dr. Akbari, a pioneer in breast cancer surgery, brings new line of treatment services to the clinic
Fairfax, Va. (November 28, 2017) —Virginia Cancer Specialists (VCS) announced that Stephanie Akbari, MD, FACS, has joined the practice. VCS, a practice in The US Oncology Network, is a premier cancer care and research center in Northern Virginia with more than 40 years of service to patients.
Dr. Akbari, the first dedicated breast surgeon in Northern Virginia, has been practicing in the area since 2001. Continually integrating new and innovative technology into treatments, Dr. Akbari established the first nationally accredited breast cancer center in the Washington, D.C., region in 2007. There, Dr. Akbari became the first in the area to become certified in breast ultrasound, incorporate partial breast irradiation, and perform office-based minimally invasive breast biopsies.
“We are delighted to have Dr. Akbari, a true leader in the field in breast cancer treatment, join our growing practice,” said Linda Anderson, executive director of VCS. “With her vision and focus on promising new advancements in breast cancer treatment, Dr. Akbari is a welcome addition to the treatment team as we seek to deliver the very best in cancer care to the Northern Virginia community.”
“It is very exciting to join Virginia Cancer Specialists,” said Dr. Akbari. “Throughout my career, I have admired VCS’s cutting-edge and integrated approach to treating cancer patients. I am thrilled to join this incredible team of professionals dedicated to providing comprehensive breast cancer care, one that has such a long-standing, strong commitment to research.”
Dr. Akbari earned her undergraduate degree from Mount Holyoke College. She received her medical training at the University of Pennsylvania and completed her residency in surgery at Harvard’s New England Deaconess Hospital. She is a fellow of the American College of Surgeons and a member of the American Society of Breast Surgeons, previously serving on committees of both organizations. Since 2005, Dr. Akbari has been recognized as one of the region’s “Top Docs” in breast surgery by Washingtonian Magazine.
About Virginia Cancer Specialists
For more than 40 years, Virginia Cancer Specialists (VCS) has contributed to the campaign against cancer and diseases of the blood. VCS has nine locations throughout Northern Virginia staffed by highly-skilled physicians, each delivering exceptional care. The practice has built a world-class treatment team of cancer specialists, as well as acquiring the very latest treatment technology to help achieve the best outcomes for patients. VCS unites medical and radiation oncologists, breast and musculoskeletal surgeons, hematologists, palliative care physicians, oncology nurse navigators, oncology infusion nurses, radiation therapists, genetic counselors, oncology pharmacists and laboratory technicians. All working together, this team designs the optimal multidisciplinary treatment program, efficiently shares knowledge, executes the treatment plan, and provides a patient-centered, consumer-friendly approach to cancer care. Patients receive the full spectrum of high-quality care necessary to treat their disease from a care team united in their effort to provide each patient with the specific personalized care they need to battle cancer. The physicians at VCS have published more than 100 abstracts and papers over the last few years and have been invited to present at national meetings. To learn more about Virginia Cancer Specialists, please visit http://www.virginiacancerspecialists.com/.
Virginia Cancer Specialists is an affiliate of The US Oncology Network (The Network). This collaboration unites VCS with more than 1,400 independent physicians dedicated to delivering value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The US Oncology Network is supported by McKesson Specialty Health, whose coordinated resources and infrastructure allow doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. VCS also participates in clinical trials through US Oncology Research, which has played a role in more than 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information, visit www.usoncology.com.
Media Contacts
Michelle Keene, Virginia Cancer Specialists
571-499-1914
Michelle.Keene@usoncology.com
Claire Crye, The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
###