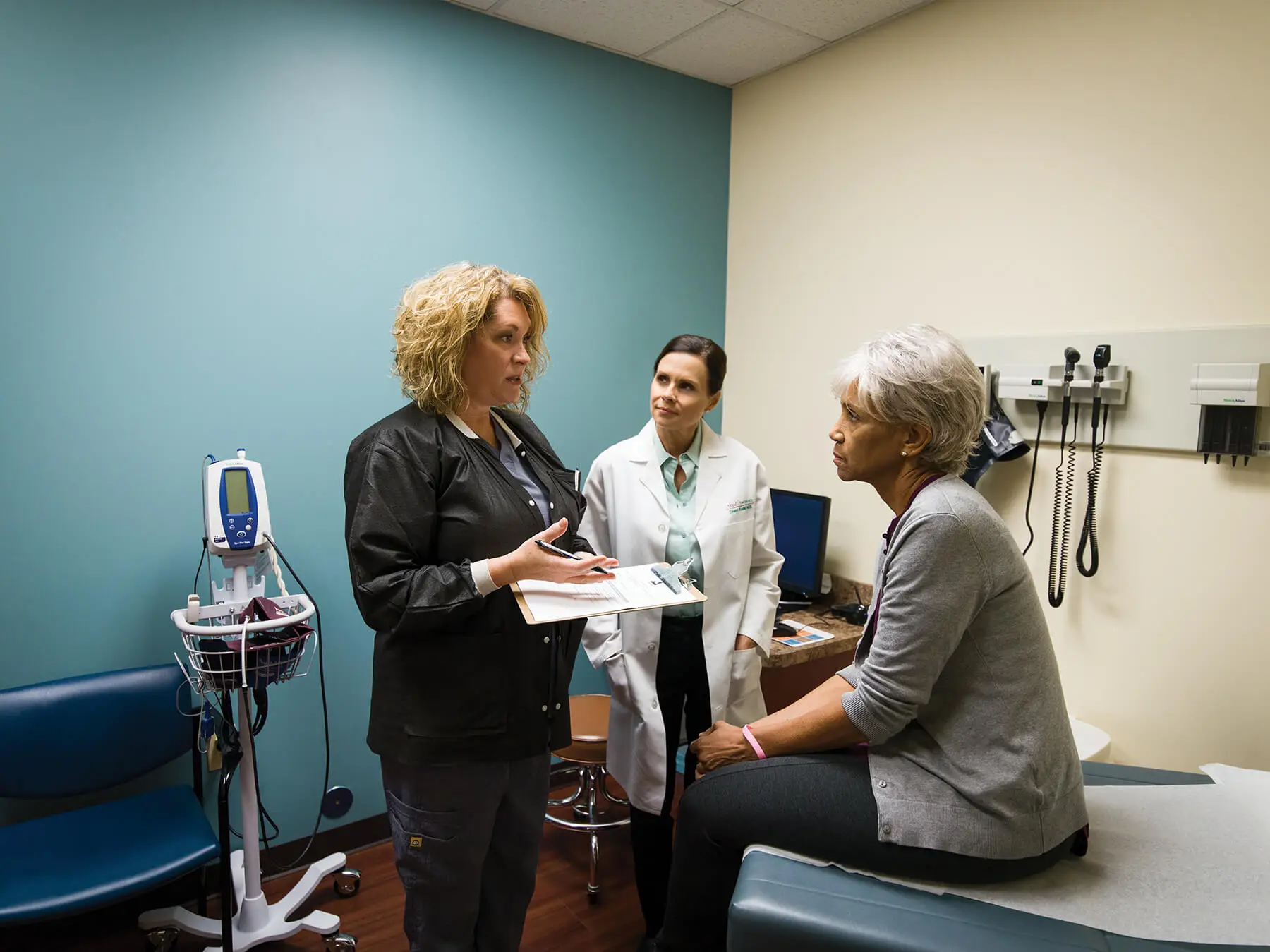
Affiliation enhances care at both practices, expanding Maryland Oncology Hematology’s services into the Baltimore–Washington Metropolitan Area
Columbia, Md. (November 3, 2017) — Maryland Oncology Hematology (MOH), a premier provider of community-based advanced cancer care services throughout the Maryland/Washington D.C. region and an affiliate of The US Oncology Network (The Network), announced that Frederick Oncology & Hematology Associates (FOHA) has joined its practice. With the addition of FOHA, Maryland Oncology Hematology broadens its service area into the Baltimore–Washington Metropolitan Area, while FOHA gains access to the many resources available from MOH and The US Oncology Network. The US Oncology Network is supported by McKesson Specialty Health, an industry leader in solutions that empower the community patient care delivery system to advance the science, technology and quality of care.
“Frederick Oncology & Hematology Associates is highly regarded in the medical community as a cancer practice that provides the very latest cutting-edge treatments to its patients, coupled with personalized, compassionate care,” said Edward J. Lee, M.D., president of MOH. “The addition of this highly-skilled group of like-minded physicians and their staff brings additional expertise and new ideas to our practice. It will also allow us to continue our mission of providing high-quality, community-based care by adding two convenient locations to serve patients.”
Frederick Oncology & Hematology Associates’ four medical oncologists, one internal medicine physician and two advanced practice providers will join MOH’s 29 physicians to offer advanced treatment options and high-quality integrated cancer care in convenient community settings across Maryland and the surrounding D.C. area. The affiliation will allow MOH to expand its comprehensive cancer care services by adding treatment locations in Frederick and Mt. Airy, Maryland.
“By uniting with Maryland Oncology Hematology, we enhance our ability to provide high-quality advanced care by accessing additional leading-edge technologies and therapies, including groundbreaking clinical trials that our patients can participate in close to home,” said Kanan H. Hudhud, M.D., medical oncologist with FOHA. “We also look forward to utilizing some of the innovative business support tools available from The Network to increase efficiencies. We are excited to join MOH and The Network, as we believe this collaboration positions us well to thrive in today’s evolving environment, both clinically and operationally.”
Maryland Oncology Hematology is affiliated with The US Oncology Network, one of the nation’s largest networks of integrated community-based oncology practices. By joining MOH, physicians from Frederick Oncology & Hematology Associates gain access to the extensive resources of The Network, such as the award winning iKnowMedSM electronic health record and the innovative Lynx Mobile® drug inventory management system, as well as Value Pathways powered by NCCN,®evidence-based treatment guidelines developed by The Network physicians working with the National Comprehensive Cancer Network. Physicians from FOHA can also participate in novel clinical trials through US Oncology Research, the renowned research arm of The Network that has played a role in approximately 70 FDA-approved cancer therapies. Additionally, FOHA physicians can interact with a large physician community through participation in committees, workshops, conferences and by accessing The Oncology Portal, where 1,400 physicians from across The Network can share best practices and collaborate on complex cases in a secure online forum.
“Since the majority of cancer care is delivered in community-based settings, it is extremely important to cancer patients and their families that practices like Frederick Oncology & Hematology Associates remain strong and viable at a time when so many independent cancer centers are closing or consolidating,” said Ella Friedman, senior vice president of Operations for The US Oncology Network. “Uniting with The Network enables practices to tap into our vast operational and clinical solutions that drive quality care while enabling providers to meet the challenges today’s value-based environment presents. We welcome Frederick Oncology & Hematology Associates into The Network, and we look forward to collaborating with these highly-skilled cancer specialists to achieve our common goals of improving access, quality and cost of cancer care.”
About Maryland Oncology Hematology
Maryland Oncology Hematology (MOH) provides comprehensive, compassionate care to people challenged by cancer and blood disorders in Maryland and the surrounding D.C. area. Founded in 1986, MOH has evolved into a world-class treatment center offering a fully integrated approach to cancer treatment. Its dedicated physicians, nurses and staff guide patients and their families through treatment options, tailoring services to each patient’s unique, individual needs. State-of-the-art infusion centers offer a full range of chemotherapy services, laboratory testing, clinical trials and support services provided by people who have an unwavering commitment to achieving the best possible result. Maryland Oncology Hematology’s 34 physicians practice in 11 locations, providing comprehensive cancer care throughout Maryland to the communities of Columbia, Silver Spring, Wheaton, Brandywine, Laurel, Clinton, Lanham, Rockville, Bethesda, Mt. Airy and Frederick.
Maryland Oncology Hematology is an affiliate of The US Oncology Network. This collaboration unites MOH with more than 1,400 independent physicians dedicated to delivering value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The US Oncology Network is supported by McKesson Specialty Health, whose coordinated resources and infrastructure allow doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Maryland Oncology Hematology also participates in clinical trials through US Oncology Research, which has played a role in approximately 70 FDA-approved cancer therapies, about one-third of all cancer therapies approved by the FDA to date. For more information, visit www.usoncology.com.
Media Contacts
Mary Beth Zaber, RN, BSN, MS
Maryland Oncology Hematology
410-964-2212
MaryBeth.Zaber@usoncology.com
www.marylandoncology.com
Claire Crye
The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
###
Organization celebrates enrolling 70,000 patients to clinical trials, contributing to more than 70 FDA cancer therapy approvals
The Woodlands, Texas (Nov. 2, 2017) — US Oncology Research is holding its 16th Annual Science Forum on November 2-3 in Dallas, an annual meeting that brings together more than 150 affiliated US Oncology Research physician researchers and their clinical teams from The US Oncology Network (The Network). The meeting features educational sessions, committee meetings and networking opportunities with others who share a passion for excellence in clinical research in the community setting. This year is especially noteworthy, as the organization will be commemorating its involvement in enrolling approximately 70,000 patients in clinical research trials that contributed to more than 70 cancer therapy approvals by the Federal Drug Administration (FDA). To date, US Oncology Research has played a role in about one-third of all oncology therapies that have been approved by the FDA.
US Oncology Research, supported by McKesson Specialty Health and The US Oncology Network, is one of the largest community-based research programs in the United States specializing in oncology clinical trials. It is comprised of approximately 1,000 physicians who care for patients in addition to conducting research.
“Enrolling more than 70,000 patients to clinical trials and contributing to more than 70 FDA approved cancer therapies are significant milestones for our organization,” said Sandy Smith, vice president, US Oncology Research. “I am extremely proud of our dedicated researchers, their highly-skilled staff and the practices within The Network that have played a pivotal role in developing these cutting-edge clinical trials. As a result, we have promising new therapies and innovative treatments that are impacting thousands of cancer patients every day.”
The Annual Science Forum brings together all US Oncology Research investigators for continuing education, presentations on scientific advancements that are driving cancer treatments, as well as in-depth discussions of best practices to achieve excellence in clinical research in the community setting. Ten disease program committees are focusing on scientific and clinical trends that are paving the way for new treatment approaches, while world-renowned experts in oncology research are presenting the latest information and perspective on such cutting-edge topics as immunotherapies and antibodies in cancer treatment, the CRISPR delivery system, and the critical role of senescence and exosomes in cancer.
Research nurse coordinators are a key focus for the meeting, since they play a crucial role in clinical trials. Approximately 70 nurse coordinators are gathering to discuss how to provide high-quality care for patients in clinical trials. They are also concentrating on leveraging technology and analytics to accelerate patient identification and enrollment, support study data collection and successfully navigate FDA audits. Discussions center on McKesson Specialty Health’s industry-leading iKnowMedSM Generation 2 electronic health record system and the company’s Practice InsightsSM performance analytics tools, as well as trial finder and clinical trial management technologies.
The meeting is also honoring Centum Club investigators and practices that have enrolled 100 patients or more to clinical trials. US Oncology Research congratulates all award winners for their significant contributions to clinical research.
“The Annual Science Forum gives our US Oncology Research Team a great opportunity to hear about the very best new science with the highest possibility of making a difference to our patients,” said Daniel Von Hoff, MD, FACP, chief scientific officer, US Oncology Research. “This year we will hear debate on the best way to harness a person’s immune system against cancer, new agents with dramatic anti-cancer responses, new techniques for early detection of cancer and breakthrough approaches to cancer therapy including CRISPR, calorie restriction and eliminating cellular senescence.”
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves 60 research sites and more than 165 locations managing about 300 active trials at any given time. Physicians in the research network have enrolled more than 70,000 patients in over 1,500 trials since inception in 1992 and have played a role in more than 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information visit www.usoncology.com/oncologists.
Media Contact
Claire Crye
281-825-9927
claire.crye@usoncology.com
###
Mobile, Ala. (September 19, 2017) – Southern Cancer Center (SCC), a premier oncology clinic in Alabama and a practice in The US Oncology Network, is the first site in the world to open enrollment for a new clinical trial for newly diagnosed kidney cancer patients.
This phase 3 clinical trial will look at various investigational combination therapies including an immune checkpoint inhibitor. This is an important trial to gain information on previously untreated advanced or metastatic renal cell carcinoma (RCC).
The varieties of cancer treatment have progressed rapidly over the last few years, such that many patients now have new treatment options available to them, including immunotherapy. By activating the body’s own immune system to identify and destroy tumor cells, immunotherapies have shown significant potential and are often becoming effective treatment options for cancer care providers and their patients. However, there is still a need for ongoing research of these investigational combinations to continue the advancement of cancer treatment.
“Southern Cancer Center is proud to offer an advanced clinical research team that works closely with the National Cancer Institute, US Oncology Research and the pharmaceutical industry to explore new treatment options and bring cutting edge clinical trial opportunities to our patients,” said Dr. Michael Meshad, medical research director for Southern Cancer Center. “We’re not just treating cancer, but helping patients find the most progressive and effective treatment possible.”
SCC is conducting this clinical trial through US Oncology Research, one of the largest research programs in the U.S. focused on oncology clinical trials. US Oncology Research has participated in about 10 studies with first-in-human therapies and 30 studies with the first U.S. or global patient enrollment.
About Southern Cancer Center
The physicians and employees of Southern Cancer Center (SCC) are dedicated to finding and providing the best care and treatment options for their patients in the communities they serve. SCC has a team approach to patient care. Taking care of oncology patients is their life’s work and passion.
Southern Cancer Center is an affiliate of The US Oncology Network (The Network). This collaboration unites SCC with more than 1,400 independent physicians dedicated to delivering value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The US Oncology Network is supported by McKesson Specialty Health, whose coordinated resources and infrastructure allow doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Southern Cancer Center also participates in clinical trials through US Oncology Research, which has played a role in approximately 70 FDA-approved cancer therapies, about one-third of all cancer therapies approved by the FDA to date. For more information about clinical trials, SCC or its research program, visit www.southerncancercenter.com or call (251) 625-6896.
Media Contacts
Mary Kathryn McMillon, SCC
251-607-5033
mk.mcmillon@southerncancercenter.com
Claire Crye, The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
###
NERLYNXTM is a new treatment option for early-stage HER2+ breast cancer patients following adjuvant trastuzumab-based therapy in the extended adjuvant setting
The Woodlands, Texas (September 12, 2017) — Early-stage breast cancer patients with HER2-positive tumors now have a promising new anti-HER2 treatment option in the extended adjuvant setting, thanks in large part to the efforts and expertise of US Oncology Research and McKesson Specialty Health. US Oncology Research is the pioneering research arm of The US Oncology Network and is supported by McKesson Specialty Health. US Oncology Research played an important pivotal role supporting the drug’s manufacturer, Puma Biotechnology, Inc., through the clinical trial, ExteNET that led to approval by the U.S. Food and Drug Administration (FDA) on July 17, 2017.
FDA approval for NERLYNX was based on the Phase III ExteNET trial, a multicenter, randomized, double-blind, placebo-controlled trial of NERLYNX following adjuvant trastuzumab treatment. Women (n=2,840) with early-stage HER2-positive breast cancer and within two years of completing adjuvant trastuzumab were randomized to receive either NERLYNX (n=1,420) or placebo (n=1,420) for one year.
The results of the ExteNET trial demonstrated that after two years of follow-up, a 34% reduction in risk of recurrence was demonstrated with NERLYNX versus placebo. Invasive disease-free survival (iDFS) was 94.2% in patients treated with NERLYNX compared with 91.9% in those receiving placebo (HR 0.66; 95% CI: 0.49, 0.90, p=0.008). NERLYNX demonstrated a 27% reduction in risk of recurrence through an exploratory analysis after five years of follow up versus placebo.
The most common adverse reactions (≥5%) were diarrhea, nausea, abdominal pain, fatigue, vomiting, rash, stomatitis, decreased appetite, muscle spasms, dyspepsia, AST or ALT increase, nail disorder, dry skin, abdominal distention, epistaxis, weight loss, and urinary tract infection. The most common adverse reaction leading to discontinuation was diarrhea, which was observed in 16.8% of NERLYNX-treated patients. Increases in liver transaminases led to drug discontinuation in 1.7% of NERLYNX-treated patients.
“Neratinib gives our patients with HER2-positive tumors another highly effective oral treatment that can reduce the risk of their cancer returning,” said Frankie Ann Holmes, MD, oncologist with Texas Oncology and member of The US Oncology Network Breast Cancer Research Committee.
Dr. Holmes was the principal investigator for US Oncology Research, whose leadership and expertise played a leading role in NERLYNX’s clinical trial that led to FDA approval. US Oncology Research enrolled 230 patients to the trial―the largest number of patients from any network―representing approximately 10% of the 2,840 trial participants across the globe from North and South America, Europe, Australia, New Zealand and Japan. NERLYNX marks the 69th FDA approval in which US Oncology Research has contributed – a significant milestone.
“Collaboration between US Oncology Research and organizations such as Puma Biotechnology, Inc. are extremely critical to the success of clinical trials such as this,” noted Sandy Smith, vice president, US Oncology Research. “Everyone involved in these trials understands the importance and significance of working together and the importance of always keeping the patient as the primary focus.”
Puma Biotechnology, Inc. also collaborated with several other divisions of McKesson Specialty Health, enabling widespread practice and patient access to this new drug. McKesson Specialty Health’s oncology pharmacy services company, Biologics, Inc., was selected to be in the limited distribution network for NERLYNX as a specialty pharmacy provider. Physicians may submit prescriptions to Biologics via phone (800-850-4306), fax (800-823-4506) or eScribe. For electronic prescribing systems, physicians may search for Biologics within their EMR system.
In-office dispensing practices who would like access to NERLYNX can place orders via McKesson Specialty Health’s Customer Center or by contacting their Customer Care team at 800-482-6700.
Puma has developed the Puma Patient Lynx support program to provide patients and healthcare providers with assistance related to questions on accessing neratinib and referrals to resources that can help with reimbursement and financial assistance. More information on the Puma Patient Lynx program can be found at WWW.NERLYNX.COM or 855-816-5421.
Indication
NERLYNX™ is a tyrosine kinase inhibitor indicated for the extended adjuvant treatment of adult patients with early stage HER2-overexpressed/amplified breast cancer, to follow adjuvant trastuzumab-based therapy.
Important Safety Information
There are possible side effects of NERLYNX. Patients must contact their doctor right away if they experience any of these symptoms. NERLYNX treatment may be stopped or the dose may be lowered if the patient experiences any of these side effects.
Diarrhea
Diarrhea is a common side effect of NERLYNX. The diarrhea may be severe, and you may get dehydrated. Your healthcare provider should prescribe the medicine loperamide for you during your first 2 cycles (56 days) of NERLYNX and then as needed. To help prevent or reduce diarrhea:
- You should start taking loperamide with your first dose of NERLYNX.
- Keep taking loperamide during the first 2 cycles (56 days) of NERLYNX treatment and then as needed. Your healthcare provider will tell you exactly how much and how often to take loperamide.
- While taking loperamide, you and your healthcare provider should try to keep the number of bowel movements that you have at 1 or 2 bowel movements each day.
- Tell your healthcare provider if you have more than 2 bowel movements in 1 day, or if you have diarrhea that does not go away.
Contact your healthcare provider right away if you have severe diarrhea or if you have diarrhea along with weakness, dizziness, or fever.
Liver Problems
Changes in liver function tests are common with NERLYNX. The patient’s doctor will do tests before starting treatment, monthly during the first 3 months, and then every 3 months as needed during treatment with NERLYNX. NERLYNX treatment may be stopped or the dose may be lowered if your liver tests show severe problems. Symptoms of liver problems may include tiredness, nausea, vomiting, pain in the right upper stomach area (abdomen), fever, rash, itching, yellowing of your skin or whites of your eyes.
Pregnancy
Patients should tell their doctor if they are planning to become pregnant, are pregnant, plan to breastfeed, or are breastfeeding. NERLYNX can harm your unborn baby. Birth control should be used while a patient is receiving NERLYNX and for at least 1 month after the last dose. If patients are exposed to NERLYNX during pregnancy, they must contact their healthcare provider right away.
Common side effects in patients treated with NERLYNX
In clinical studies, the most common side effects seen in patients taking NERLYNX were diarrhea, nausea, abdominal pain, fatigue, vomiting, rash, stomatitis (dry or inflamed mouth, or mouth sores), decreased appetite, muscle spasms, dyspepsia, changes in liver blood tests results, nail problems, dry skin, abdominal distention, weight loss, and urinary tract infection.
Patients should tell their doctor right away if they are experiencing any side effects. Report side effects to the FDA at 1-800-FDA-1088 or http://www.FDA.gov/medwatch. Patients and caregivers may also report side effects to Puma Biotechnology at 1-844-NERLYNX (1-844-637-5969).
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves 60 research sites and 165 locations managing about 350 active trials at any given time. Physicians in the research network have enrolled more than 68,000 patients in over 1,500 trials since inception in 1992 and have played a role in approximately 70 FDA-approved cancer therapies, about one-third of all cancer therapies approved by the FDA to date. For more information visit www.usoncology.com/physicians/clinical-trials.
About McKesson Specialty Health
McKesson Specialty Health, a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, manufacturer and payer solutions, McKesson Specialty Health focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit www.mckessonspecialtyhealth.com.
Media Contact
Claire Crye
281-825-9927
claire.crye@mckesson.com
###
Eligible Patients to be Directed to Epizyme’s Ongoing Phase 2 Clinical Trial of Tazemetostat in Follicular Lymphoma and Diffuse Large B-cell Lymphoma
THE WOODLANDS, Texas and CAMBRIDGE, Mass. (August 1, 2017) – Epizyme, Inc. (NASDAQ: EPZM), a clinical-stage biopharmaceutical company creating novel epigenetic therapies, and US Oncology Research, one of the nation’s largest networks of independent, community-based oncology practices dedicated to advancing high-quality treatments through clinical trials, today announced a collaboration to screen and identify relapsed or refractory follicular lymphoma (FL) and diffuse large B-cell lymphoma (DLBCL) patients with EZH2 mutations. Once identified, eligible candidates will be directed to Epizyme’s ongoing Phase 2 clinical trial of tazemetostat, the Company’s first-in-class EZH2 inhibitor, as a single-agent treatment for relapsed or refractory patients with FL or DLBCL.
“Relapsed follicular lymphoma and DLBCL are difficult diseases for which there is generally poor prognosis and limited treatment options for patients,” said Michael Seiden, M.D., Ph.D. chief medical officer, US Oncology Research. “Physicians affiliated with US Oncology Research strive to provide access to the treatment strategies that are best suited to each patient. Given the encouraging clinical activity and favorable safety profile demonstrated by tazemetostat in these patient populations, US Oncology Research is pleased to work with Epizyme to offer this screening process so that appropriate patients being treated by an affiliated physician can be promptly directed to a clinical trial evaluating an investigational therapy that is targeted to their cancer.”
Under the collaboration, US Oncology Research will implement a separate screening protocol in 68 locations in the U.S. to identify relapsed or refractory FL and DLBCL patients with tumors bearing EZH2 mutations who may be candidates for enrollment in Epizyme’s ongoing Phase 2 clinical trial. US Oncology Research will direct identified patients to the tazemetostat Phase 2 clinical trial for protocol screening and potential enrollment into the trial. Sites began screening patients in July 2017.
“We are pleased that US Oncology Research, a program recognized for tremendous success in oncology clinical trials and for providing patients with access to novel treatments, is joining our effort to develop a targeted treatment for lymphoma patients with EZH2 mutations,” said Peter Ho, chief medical officer of Epizyme. “This collaboration significantly expands our clinical trial footprint within the United States and is expected to further enhance our enrollment of patients whose tumors harbor an EZH2 mutation for our ongoing Phase 2 study of tazemetostat. We believe that tazemetostat has the potential to play a very important role in the targeted treatment of these patients in the future.”
About Follicular Lymphoma (FL) and Diffuse Large B-Cell Lymphoma (DLBCL)
FL, an indolent form of non-Hodgkin lymphoma (NHL), is considered to be incurable with existing treatments and is characterized by cycles of relapse that become increasingly difficult to treat with each disease progression. Approximately 25,000 patients in the U.S. and major European countries are diagnosed with FL every year1, of which 15 to 20 percent are estimated to have an EZH2 mutation. There are no approved treatments indicated for patients with FL with an EZH2 mutation. In April 2017, the FDA granted Fast Track designation to tazemetostat for FL regardless of EZH2 mutational status.
DLBCL is an aggressive form of NHL that, once diagnosed, typically requires immediate treatment. Approximately 45,000 patients are diagnosed with DLBCL in the U.S. and major European countries every year2. Among patients with germinal center DLBCL, an estimated 15 to 20 percent have an EZH2 mutation. Forty to 50 percent of patients will relapse on their first-line treatment, which is most commonly the chemotherapy regimen R-CHOP, and there are few treatment options for patients who relapse or become refractory to chemotherapy. In November 2016, the FDA granted Fast Track designation to tazemetostat for DLBCL with EZH2 mutations.
About the Tazemetostat Clinical Trial Program
Tazemetostat, a first-in-class EZH2 inhibitor, is currently being studied in ongoing Phase 2 programs in both follicular lymphoma (FL) and diffuse large B-cell lymphoma (DLBCL) forms of non-Hodgkin lymphoma; certain molecularly defined solid tumors, including epithelioid sarcoma and other INI1-negative tumors; and mesothelioma, as well as in combination studies in DLBCL. Tazemetostat has been granted Fast Track designation by the U.S. Food and Drug Administration for FL regardless of EZH2 mutation and for DLBCL with EZH2-activating mutations, as well as Orphan Drug designation for soft tissue sarcoma and malignant rhabdoid tumors.
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves 60 research sites and 165 locations managing about 350 active trials at any given time. Physicians in the research network have enrolled nearly 68,000 patients in over 1,500 trials since inception in 1992 and have played a role in nearly 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information visit https://www.usoncology.com/physicians/clinical-trials.
About Epizyme, Inc.
Epizyme, Inc. is a clinical-stage biopharmaceutical company committed to rewriting cancer treatment through novel epigenetic medicines. Epizyme is broadly developing its lead product candidate, tazemetostat, a first-in-class EZH2 inhibitor, with studies underway in both solid tumors and hematological malignancies, as a monotherapy and combination therapy and in relapsed and front-line disease. Using the Company’s proprietary platform, Epizyme has pioneered the identification and development of small molecule inhibitors of chromatin modifying proteins (CMPs), such as tazemetostat. CMPs are part of the system of gene regulation, referred to as epigenetics, that controls gene expression. Genetic alterations can result in changes to the activity of CMPs, which can allow cancer cells to grow and proliferate. By focusing on the genetic drivers of cancers, Epizyme’s science seeks to match targeted medicines with the specific patients that need it. For more information, visit www.epizyme.com and connect with us on Twitter at @EpizymeRx.
Cautionary Note on Forward-Looking Statements
Any statements in this press release about future expectations, plans and prospects for Epizyme, Inc. (the Company) and other statements containing the words “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “target,” “potential,” “will,” “would,” “could,” “should,” “continue,” and similar expressions, constitute forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: uncertainties inherent in the initiation of future clinical studies and in the availability and timing of data from ongoing clinical studies; whether interim results from a clinical trial will be predictive of the final results of the trial; whether results from preclinical studies or earlier clinical studies will be predictive of the results of future trials; whether the rate of enrollment in the Company’s trials will be increased meaningfully or at all; whether results from clinical studies will warrant meetings with regulatory authorities or submissions for regulatory approval; expectations for regulatory approvals to conduct trials or to market products; whether the Company’s cash resources will be sufficient to fund the Company’s foreseeable and unforeseeable operating expenses and capital expenditure requirements; other matters that could affect the availability or commercial potential of the Company’s therapeutic candidates; and other factors discussed in the “Risk Factors” section of the Company’s most recent Form 10-Q filed with the SEC and in the Company’s other filings from time to time with the SEC. In addition, the forward-looking statements included in this press release represent the Company’s views as of the date hereof and should not be relied upon as representing the Company’s views as of any date subsequent to the date hereof. The Company anticipates that subsequent events and developments will cause the Company’s views to change. However, while the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so.
1 Decision Resources, 2017
2 Decision Resources, 2017
Contacts
Claire Crye, US Oncology Research
Claire.Crye@usoncology.com
281-825-9927
Cheya Pope, Epizyme, Inc.
media@epizyme.com
617-229-7561
Monique Allaire, THRUST IR
monique@thrustir.com
(617) 895-9511
###
Intensive workshop provided resources, expertise to help practices succeed with the Oncology Care Model, MIPS and other alternative payment programs
The Woodlands, Texas (June 23, 2017) — The US Oncology Network (The Network) hosted its second annual boot camp last month in Dallas, focusing on various value-based care delivery and reimbursement models. The intensive two-day workshop brought together more than 100 key stakeholders and thought leaders in value-based care, including physicians, clinical staff, quality leads, and executive leaders from The US Oncology Network affiliated practices across the nation. Participants shared best practices and lessons learned from participation in emerging payment models, with a focus on the Center for Medicare and Medicaid Innovation’s Oncology Care Model, the Merit-Based Incentive Payment System (MIPS) and other alternative value-based programs. The special seminar was a collaborative effort of The US Oncology Network and McKesson Specialty Health.
“Value-based care is no longer an initiative of the future, it is here today,” said Marcus Neubauer, MD, vice president and medical director, Payer and Clinical Services, McKesson Specialty Health and The US Oncology Network. “Our boot camps enable us to collaborate and share insights and knowledge that The Network affiliated practices have gained. As practices undergo the major transformation required to accommodate this new way of delivering care, we will continue to develop innovative solutions that enable them to succeed in this transition while keeping patient care their number one priority.”
Seminar attendees heard presentations from their peers and McKesson Specialty Health thought leaders on topics aimed at providing practical information to help practices prosper―including how to lead change while managing daily operations and interactive discussions on how care team huddles can raise quality of care and staff engagement. Speakers gave updates on technology enhancements and practice tools, legislative activities and the future political outlook, as well as an overview of upcoming developments in The Network. Breakout sessions also provided attendees with more intimate collaboration opportunities, focusing on such critical issues as managing care for patients on oral therapies, establishing care team centered patient navigation, 24/7 care, enhanced patient services, data sharing and goal setting, and more.
“The boot camp illustrates our strong commitment to support community-based practices as they embrace value-based care initiatives such as the Oncology Care Model and MIPS,” said Diana Verrilli, senior vice president, Payer and Practice Management Solutions, McKesson Specialty Health. “As industry leaders in value-based care, we are dedicated to providing The Network with a wide range of comprehensive, proven solutions that support the critical components necessary to build and maintain a strong viable practice in the new value-based environment.”
The US Oncology Network, supported by McKesson Specialty Health, is leading the way in value-based care, developing the innovative technologies, resources and proficiency required to empower independent oncology practices to thrive in the changing healthcare landscape. With more than 1,400 physicians delivering care in over 400 community-based treatment sites, The Network has substantial experience with alternative payment models and is continually striving to enhance its solutions that address these new care delivery and reimbursement programs. The boot camp is just one of many opportunities The US Oncology Network provides for its affiliated practices to collaborate and come together to share knowledge, resources and expertise, enabling all members of The Network to succeed together.
About The US Oncology Network
Every day, The US Oncology Network helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About McKesson Specialty Health
McKesson Specialty Health, a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, manufacturer and payer solutions, McKesson Specialty Health focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit mckessonspecialtyhealth.com.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@mckesson.com
# # #
Expanded agreement enhances Value Pathways, providing physicians access to the most recent evidence available for five additional disease states
The Woodlands, Texas (June 14, 2017)— McKesson and The US Oncology Network (The Network) have expanded their agreement with the National Comprehensive Cancer Network® (NCCN®), adding five additional disease states to Value Pathways powered by NCCN. Value Pathways powered by NCCN are evidence-based treatment recommendations for oncology delivered through McKesson’s industry-leading clinical quality and regimen support system, Clear Value PlusSM. New cancers added include bladder, esophageal, gastric, kidney, and myelodysplastic syndromes (MDS), making Clear Value Plus even more robust. The point-of-care support tool now provides detailed information on 27 different disease states. The expanded relationship also calls for the addition of up to 15 more disease states in the next three years and more in the future, ensuring that the more than 1,500 providers utilizing Clear Value Plus will have quick and easy access to the very latest information and research on each cancer.
“Our extended agreement with NCCN is part of our ongoing commitment to provide Clear Value Plus users access to the most recent evidence available on various cancers, enabling them to deliver high-quality care,” said Marcus Neubauer, MD, vice president and medical director, Payer and Clinical Services, McKesson and The US Oncology Network. “The agreement also demonstrates our strong relationship with NCCN, which will only strengthen as we continue to collaborate in the future to advance cancer care by developing Value Pathways for more diseases and disciplines.”
Both Value Pathways powered by NCCN and NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) are integrated into the clinical workflow and Electronic Health Records by Clear Value Plus. NCCN Guidelines are the most comprehensive and most frequently updated clinical practice guidelines available in any area of medicine, and they are recognized across the industry as the gold standard in cancer care. Clear Value Plus is the only technology that includes the full logic of NCCN Guidelines®. The rigorous logic of these Guidelines is embedded as the foundational evidence source within Clear Value Plus, supplemented by the collaboratively-developed Value Pathways. Grounded in clinical quality and evidence-based medicine, Value Pathways powered by NCCN helps ensure patients receive high-quality care that offers the best opportunity for positive outcomes, while also recognizing the importance of value in determining appropriate treatment options.
“NCCN is pleased to expand our collaboration with McKesson Specialty Health and further enhance Value Pathways powered by NCCN,” said Robert W. Carlson, MD, chief executive officer, NCCN. “In supporting this expanded agreement, both NCCN and McKesson recognize the value of having evidence-based NCCN Guidelines recommendations at the point of care facilitated through technology to achieve the best outcomes for patients with cancer.”
Value Pathways powered by NCCN and Clear Value Plus enable practices to succeed in today’s evolving value-based environment by driving high-quality care and confirming that selected therapies are consistent with nationally recognized clinical guidelines as required by value-based programs, including the Oncology Care Model. In addition to tracking pathways adherence, Clear Value Plus also supports value-based care by integrating financial information, benchmarking and providing up to date reporting on key performance metrics, such as regimen utilization history and program participation for individual providers, a single site or across an entire practice. Additionally, the increased financial transparency provided by Value Pathways and Clear Value Plus allows evaluation of the cost-effectiveness of different treatment options with similar outcomes, an important element in value-based care.
“Value Pathways powered by NCCN and Clear Value Plus enable us to deliver high-quality, evidence-based care that gives our patients the best chance for a positive result and a good quality of life while providing high value to both patients and payers,” said Debra Patt, MD, MPH, MBA, vice president of Public Policy and Academic Affairs for Texas Oncology, an affiliate of The US Oncology Network. Dr. Patt recently authored a study on the topic of clinical decision support presented at ASCO Quality titled, “Measuring quality in cancer care: Utilizing a clinical decision support system to assess pathway adherence.”
Dr. Patt concluded, “The expanded agreement with NCCN strengthens our ability to deliver excellent care by providing access to the most recent evidence available for five more disease states with many more to come, impacting patient care today and into the future.”
The National Comprehensive Cancer Network® (NCCN®)
The National Comprehensive Cancer Network® (NCCN®), a not-for-profit alliance of 27 of the world’s leading cancer centers devoted to patient care, research, and education, is dedicated to improving the quality, effectiveness, and efficiency of cancer care so that patients can live better lives. Through the leadership and expertise of clinical professionals at NCCN Member Institutions, NCCN develops resources that present valuable information to the numerous stakeholders in the health care delivery system. As the arbiter of high-quality cancer care, NCCN promotes the importance of continuous quality improvement and recognizes the significance of creating clinical practice guidelines appropriate for use by patients, clinicians, and other health care decision-makers. Clinicians, visit NCCN.org. Patients and caregivers, visit NCCN.org/patients. Media, visit NCCN.org/news.
About The US Oncology Network
Every day, The US Oncology Network (The Network) helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About McKesson
McKesson Corporation, currently ranked 6th on the FORTUNE 500, is a global leader in healthcare supply chain management solutions, retail pharmacy, community oncology and specialty care, and healthcare information technology. McKesson partners with pharmaceutical manufacturers, providers, pharmacies, governments and other organizations in healthcare to help provide the right medicines, medical products and healthcare services to the right patients at the right time, safely and cost-effectively. United by our ICARE shared principles, our employees work every day to innovate and deliver opportunities that make our customers and partners more successful — all for the better health of patients. McKesson has been named the “Most Admired Company” in the healthcare wholesaler category by FORTUNE, a “Best Place to Work” by the Human Rights Campaign Foundation, and a top military-friendly company by Military Friendly. For more information, visit www.mckesson.com.
Media Contacts
Claire Crye
McKesson
281-825-9927
claire.crye@mckesson.com
Katie Kiley Brown
National Comprehensive Cancer Network (NCCN)
215-690-0238
brown@nccn.org
###
New facility developed by Ryan Companies US, Inc. offers leading-edge cancer care to growing East Valley region
Tempe, Ariz. (June 13, 2017) — Arizona Oncology’s new state-of-the-art cancer treatment center, located at the Arizona State University Research Park (ASURP), is now open and offering the latest cancer treatment services available to patients in Phoenix and the East Valley. The new center, developed and built by Ryan Companies US, Inc., offers integrated, high-quality care to diagnose and treat cancer. Patient services include medical, gynecologic and radiation oncology, hematology, advanced diagnostics including PET/CT imaging, supportive care services, and clinical trials. The practice is also offering the latest treatment options for patients, including immuno-oncology, bio markers, and targeted therapies.
Arizona Oncology, a practice in The US Oncology Network, is one of the largest medical groups in Arizona devoted exclusively to cancer care. This is the practice’s twenty-first location in the Phoenix area and their first location in Tempe – the company is expanding its services to Elliot Road and Loop 101 to provide more patients with convenient and advanced cancer care closer to home.
“We have been treating patients for more than 20 years in the valley and understand the importance of offering specialized treatment close to home,” said Snehal Bhoola, MD, gynecologic oncologist, Arizona Oncology. “We pride ourselves on our patient-centered approach, which allows us to offer high-quality care.”
This nearly 22,000 square-foot building is a hub for cancer care and the only one of its kind in the East Valley. The center will support six full-time oncologists, including two gynecologic oncologists, three medical oncologists, and one radiation oncologist. The facility will have two treatment vaults for radiation, a room dedicated to PET-CT scanning, 35 infusion chairs with personal tablets, 19 exam rooms, a covered patio with peaceful waterfalls, and a nursing station with semi-private scheduling areas. Additionally, the center will offer Vital Beam linear accelerator and High-Dose-Rate (HDR) Brachytherapy treatment options, in-office dispensing, cancer genetic counseling, and support services that will help patients prevent or cope with a cancer diagnosis. Social workers, nurse navigators, and patient benefit representatives will also be available to patients.
Butler Design Group designed the shell of the building, and David B. Howell Architecture & Design was tasked with designing the interior, which is filled with cutting-edge technology and comforting aesthetic features including the pharmacy’s isolated independent air system, the serene visual ceiling displays in the radiation vaults and a healing garden.
“The ASU Research Park is a premiere location for innovation by leading companies, which benefit from the opportunity to collaborate with ASU intellectual assets in a world-class business park setting,” said Morgan R. Olsen, ASU Research Park Board President and ASU Executive Vice President, Treasurer and Chief Financial Officer. “We are thrilled to welcome Arizona Oncology, a practice committed to advancing treatment modalities to help defeat cancer in its many forms. Arizona Oncology also will further Valley economic development, adding to the ASU Research Park’s contribution of more than 6,000 jobs at over 50 companies.”
This free-standing treatment and research center was constructed by Ryan Companies in just nine months. This is Ryan’s tenth build-to-suit project developed and constructed in the ASURP.
“Bringing Arizona Oncology’s expertise and innovative technology to the prestigious ASURP is a tremendous addition for East Valley residents needing high-quality care,” said Molly Ryan Carson, vice president of Development at Ryan. “Our team worked diligently to ensure maximum accuracy during construction on this vital healthcare clinic.”
Arizona Oncology is now accepting patients at the new location. To schedule an appointment, visit AOAppt.com, or for more information, call 888-972-CURE or visit http://www.arizonaoncology.com/.
About Arizona Oncology
Arizona Oncology is one of the largest medical groups in Arizona. With more than 75 practicing physicians devoted exclusively to providing comprehensive, compassionate and high-quality cancer care, Arizona Oncology specializes in Medical, Gynecologic, and Radiation Oncology, Hematology, Stem Cell Transplant, Research, Genetic Risk Assessment and patient ancillary programs. The physicians and their staff treat patients in many communities throughout the state including: Deer Valley, East Valley (Tempe), Estrella, Flagstaff, Glendale, Green Valley, Nogales, Peoria, Phoenix, Prescott Valley, Safford, Scottsdale, Sedona, and Tucson.
Arizona Oncology believes it is beneficial to provide cancer therapies in a community setting, close to patients’ homes and support systems. The physicians are supported by a talented clinical team sensitive to the needs of cancer patients and their caregivers. For more information, visit http://www.arizonaoncology.com/.
Arizona Oncology is an affiliate of The US Oncology Network (The Network). This collaboration unites Arizona Oncology with more than 1,400 independent physicians dedicated to delivering value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The US Oncology Network is supported by McKesson Specialty Health, whose coordinated resources and infrastructure allow doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Arizona Oncology also participates in clinical trials through US Oncology Research, which has played a role in nearly 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information, visit www.usoncology.com.
About Ryan Companies US, Inc.
Founded in 1938, Ryan Companies offers comprehensive commercial real estate services as a national developer, architect, capital investment consultant, builder and real estate manager with a focus on bringing lasting value to its customers and the communities in which it works. Ryan market depth includes retail, industrial, healthcare and senior living. Ryan development and corporate build-to-suit work spans a wide range of product types including office, mixed-use, hospitality, multifamily housing and mission critical facilities. Ryan has nearly 1,300 employees in 13 offices and has completed projects in 38 states. For more information, visit ryancompanies.com
About Arizona State University Research Park (ASURP)
The ASU Research Park is located on the site of a former experimental farm and was acquired with funds from the Arizona State College Foundation (now ASU Foundation) in 1956. Founded in 1984, the ASU Research Park represents ASU’s commitment to technology transfer through university-industry research. Located in the Valley’s high technology hub, the park is comprised of 320 acres and offers a strategic location with access to the southeast valley high tech employment pool. For further information, please see www.asuresearchpark.com/.
About Arizona State University
Arizona State University is a New American University – a major public educational institution, a premier research center and a leader in innovation. Our vision is described by our three core principles: excellence in scholarship, access to education, and impact in our global community. As a New American University, ASU is intellectually vibrant, socially conscious and globally engaged. For more information, please visit www.asu.edu.
Media Contact
Amy Magaziner, Arizona Oncology
480-789-2986
Amy.Magaziner@usoncology.com
###
New clinic enhances world-class research program by strengthening Phase I capabilities
Fairfax, Va. (June 8, 2017) — There is good news today for patients in Northern Virginia who are battling cancer, as well as those who may face the disease in the future. Virginia Cancer Specialists (VCS), Northern Virginia’s premier cancer care and research center with more than 40 years of service to patients and a practice in The US Oncology Network, has opened a new state-of-the-art research clinic in Fairfax, Virginia dedicated exclusively to Phase I clinical trials. A ribbon cutting ceremony was held May 24 to officially mark the opening.
Phase I studies are the earliest stage of drug development and the most advanced trials conducted in the cancer community. They are usually only available at large academic research centers, and very few community-based cancer practices like VCS can qualify as a Phase I provider. While VCS has been conducting Phase I trials since 2013, the new dedicated facility greatly enhances the practice’s ability to offer these novel early-stage studies.
“We are very excited and proud to open our new Phase I clinic, as research is a vital part of our mission to provide patients access to advanced cancer treatments in the community where they live and work,” said Alex Spira, MD, PhD, FACP, medical oncologist and director of the VCS Research Institute and Phase I Trial Program. “This new clinic not only allows us to better meet the growing demand in our community for these groundbreaking trials, it also enables us to lead the way in research focusing on promising new advancements, such as immunotherapy.”
Virginia Cancer Specialists has access to hundreds of innovative clinical trials, including a vast number of Phase I studies, through its affiliation with The US Oncology Network, the largest network of its kind comprised of more than 1,400 community-based physicians and a leader in cancer research and treatment. Many practices in The US Oncology Network conduct clinical research through US Oncology Research, which manages nearly 400 active trials across the country at any given time and has contributed to nearly 70 FDA-approved cancer therapies.
“Virginia Cancer Specialists has a long-standing, strong commitment to research, and the practice currently offers more Phase I clinical trials than any other facility in Northern Virginia,” noted Karin Choquette, MSN, RN, CCRC, clinical research manager, VCS. “Our new Phase I clinic strengthens our research leadership position in the region while giving patients access to a vast number of emerging treatment options that are not yet FDA approved.”
The new 4,000 square foot clinic incorporates the very latest design concepts to support individualized patient comfort, serenity and healing, while enabling communication and bonding among patients. Protocols for Phase I trials are demanding, and often require patients to be on site for up to 12 hours. On call services with a specialized nurse is provided 24/7 to closely monitor patients. The facility has four treatment chairs with special heating and massage features to provide a high level of comfort. Each treatment area can be curtained off to create the patient’s own private space. Family members and caregivers are welcome, as their presence provides encouragement and helps support an optimal outcome.
The facility is staffed by a dedicated team of specialized experts highly trained in clinical research, including physicians, data coordinators, nurses and clinical research coordinators. Pharmacists are also an integral part of the team, ensuring that exacting standards are met.
“Patients who participate in Phase I trials have usually exhausted all other treatment options, but they still want to fight their cancer,” explained Anita Karra, RN, a VCS Phase I nurse. “Most people battling cancer at this stage must travel great distances for a Phase I trial, so our patients are very thankful we offer these novel studies that enable them to stay in their community where they have the support of family and friends.”
The new Phase I clinic also brings VCS one step closer on its journey to become an Immuno-Oncology Center of Excellence, a long-term goal of the Research Institute. Immunotherapy is an area of great interest throughout the cancer community and especially within US Oncology Research, where immuno-oncology (I-O) trials comprise approximately one-third of all studies offered. New I-O therapies have high efficacy rates, great tolerability and manageable toxicities, with some even demonstrating durable responses after treatment ends.
“Immunotherapy is the greatest advancement we have seen in cancer research in several decades,” said Dr. Spira. “Our Phase I clinic will enable us to concentrate on investigational I-O trials, allowing us to gain valuable expertise and insight we can share with the entire medical community while providing patients access to these promising new therapies.”
About Virginia Cancer Specialists
For more than 40 years, Virginia Cancer Specialists (VCS) has contributed to the campaign against cancer and diseases of the blood. VCS has nine locations throughout Northern Virginia staffed by highly-skilled physicians, each delivering exceptional care. The practice has built a world-class treatment team of cancer specialists, as well as acquiring the very latest treatment technology to help achieve the best outcomes for patients. VCS unites medical, radiation and orthopedic oncologists, hematologists, oncology nurse navigators, oncology infusion nurses, radiation therapists, genetic counselors, oncology pharmacists and laboratory technicians, all working together as a team to design the optimal multidisciplinary treatment program, efficiently sharing knowledge, executing the treatment plan, and providing a patient-centered, consumer-friendly approach to cancer care. Patients receive the full spectrum of high-quality care necessary to treat their disease from a care team united in their effort to provide each patient with the specific personalized care they need to battle cancer. The physicians at VCS have published more than 100 abstracts and papers over the last few years, and have been invited to present at national meetings. To learn more about Virginia Cancer Specialists, please visit http://www.virginiacancerspecialists.com/.
Virginia Cancer Specialists is an affiliate of The US Oncology Network (The Network). This collaboration unites VCS with more than 1,400 independent physicians dedicated to delivering value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The US Oncology Network is supported by McKesson Specialty Health, whose coordinated resources and infrastructure allow doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. VCS also participates in clinical trials through US Oncology Research, which has played a role in nearly 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information, visit www.usoncology.com.
Media Contact
Claire Crye, The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
###
Approximately 40 abstracts accepted for presentation, highlighting the importance of community-based cancer research in developing new and novel cancer fighting therapies in areas like hematology, breast cancer, and immuno-oncology
Debra Patt, MD, MBA, MPH, to speak during an Educational Session on creating an informatics-driven oncology practice
The Woodlands, Texas (May 31, 2017) – Community oncologists affiliated with The US Oncology Network (The Network) and US Oncology Research will showcase findings from approximately 40 data presentations at the 53rd Annual Meeting of the American Society of Clinical Oncology (ASCO), held June 2-6, 2017 at McCormick Place in Chicago. These data presentations cover novel approaches to numerous types of cancer specialties including hematology, gastrointestinal, breast cancer and promising immunotherapy research. In addition, experts from The Network will be sharing expert perspectives during ASCO. In particular, Debra Patt, MD, MBA, MPH, will participate in an expert panel on healthcare information technology titled, “More Medicine, Fewer Clicks: How Informatics Can Actually Help Your Practice.“
Each year, more than 1.5 million people in the United States are diagnosed with cancer, and research shows that earlier detection, diagnosis, and treatment increase patients’ chances of long-term survival.[i] In 2015, the Biden Moonshot Cancer program was launched to achieve 10 years of progress, in preventing, diagnosing and treating cancer, in five years. Although the healthcare landscape is changing and evolving, it’s imperative that the vitality of community cancer care is preserved, as many patients rely on resources close to their families and homes.
“Inspired by the Biden Moonshot Cancer program’s commitment to developing new cancer technologies, community oncologists continue to make significant contributions in shaping the future of cancer care,” said Michael V. Seiden, MD, PhD, chief medical officer for McKesson Specialty Health and The US Oncology Network. “The US Oncology Network is comprised of more than 1,400 independent physicians dedicated to delivering value-based, integrated care for patients – close to home. Many of those practices conduct research through US Oncology Research, one of the largest community-based oncology research programs in the United States. Over the past 25 years, the organization has enrolled more than 66,000 patients into clinical trials and helped bring nearly 70 therapeutic treatments to market through participation in pivotal registration trials. These efforts underscore one way community oncologists are influencing how the war against cancer is waged.”
Two US Oncology Research affiliated investigators have been selected to give oral presentations on Saturday, June 3 from 3:00 PM to 6:00 PM Central Daylight Time (CDT) in S100bc, as part of a larger session focused on Hematologic Malignancies—Lymphoma and Chronic Lymphocytic Leukemia.
- David J. Andorsky, M.D., oncologist and hematologist with Rocky Mountain Cancer Centers, an affiliate of US Oncology Research, will present a study he co-authored titled, “Phase IIIb randomized study of lenalidomide plus rituximab (R2) followed by maintenance in relapsed/refractory NHL: Analysis of patients with double-refractory or early relapsed follicular lymphoma (FL).”
- Jeff Sharman, M.D., director of Research at Willamette Valley Cancer Institute and medical director of Hematology Research for The US Oncology Network, will present a study he co-authored titled, “Ublituximab and ibrutinib for previously treated genetically high-risk chronic lymphocytic leukemia: Results of the GENUINE phase 3 study.”
“Despite recent advances in the treatment of follicular lymphoma, there remain groups of patients with this disease who have poor outcomes,” said Dr. Andorsky. “Early results from the MAGNIFY trial of rituximab plus lenalidomide in high-risk patients with follicular lymphoma found that patients with poor outcomes include those whose disease relapses early (within 2 years of initial treatment) and who are refractory to both rituximab and alkylating agents. I’m committed to helping develop novel therapies that improve the quality of care for these patients.”
Dr. Sharman said, “Research is key to the treatment and cure of hematologic malignancies. We must continue to accelerate discoveries into meaningful clinical trials to improve patient outcomes. Data from the GENUINE trial show that high-risk chronic lymphocytic leukemia patients who received a combination of ublituximab (UTX) and ibrutinib (IB) demonstrated a superior response rate compared to those who received IB alone without additional clinically significant toxicity. These results are a testament to the treatment advances being made, but there is still work to be done. We must think through our patients’ journeys, from start to finish.”
Affiliated oncologists are also participating in pre-conference and educational sessions by sharing insights and specialized expertise to help fellow oncologists continue to improve patient outcomes while advancing their practices. Oncology practices face complex decisions about the health technology landscape that help improve care and efficiencies. Selecting technologies that address the unique needs of an oncology practice and allow for improved identification of research and care options are essential. McKesson Specialty Health, which supports The US Oncology Network and US Oncology Research, provides coordinated resources and infrastructure that allow doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices.
“Big data and technology are playing a large role in improving cancer care, by giving physicians access to key information on proposed treatment plans and allowing them to develop individualized care plans for their patients,” said Dr. Patt, vice president of Public Policy and Academic Affairs at Texas Oncology and chair of The US Oncology Network Pathways Taskforce for Breast Cancer. “Appropriate use of big data can enhance efficiencies, improve outcomes, advance research, and reduce healthcare costs for patients and practices.”
A complete list of US Oncology Research affiliated studies and educational programs being featured at ASCO can be found here.
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves 60 research sites and approximately 170 locations managing nearly 400 active trials at any given time. Physicians in the research network have enrolled more than 66,000 patients in over 1,500 trials since inception in 1992 and have played a role in nearly 70 FDA-approved cancer therapies, approximately one-third of cancer therapies approved by the FDA to date. For more information visit https://www.usoncology.com/physicians/clinical-trials. To find clinical trials available through US Oncology Research visit http://trialfinder.usoncology.com.
About The US Oncology Network
Every day, The US Oncology Network (The Network) helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About McKesson Specialty Health
McKesson Specialty Health, a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, manufacturer and payer solutions, McKesson Specialty Health focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit www.mckessonspecialtyhealth.com.
Media Contacts
Claire Crye
281-825-9927
Claire.Crye@mckesson.com
Edie DeVine
209-814-9564
Edie.DeVine@gcihealth.com
###
[i] Center for Disease Control and Prevention. Cancer – Preventing One of the Nation’s Leading Causes of Death at a Glance 2016. October 2016. Accessed May 17, 2017 at https://www.cdc.gov/chronicdisease/resources/publications/aag/dcpc.htm
The Woodlands, Texas (May 12, 2017) — McKesson Corporation has played a significant role in the recent approval of ZEJULATM (niraparib). Approximately ten practices affiliated with US Oncology Research, one of the largest community-based oncology research programs in the United States supported by McKesson, participated in the clinical trial that led to approval by the U.S. Food and Drug Administration (FDA) on March 27, 2017. ZEJULA marks the 66th FDA approval in which US Oncology Research has contributed.
ZEJULA is a poly ADP-ribose polymerase (PARP) inhibitor, for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in complete or partial response to platinum-based chemotherapy. The American Cancer Society estimates that more than 22,000 women will be diagnosed with ovarian cancer in 2017. Historically, there have been few treatment options for women with advanced ovarian cancer.
Clinical trial results demonstrated that treatment with ZEJULA reduced the risk of disease progression or death by 74% in patients with germline BRCA mutations (HR 0.26) and by 55% in patients without germline BRCA mutations (HR 0.45).
“The FDA approval for ZEJULA differs from the approval of the other 2 PARP inhibitors in that it is indicated in “all comers”. A unique molecular signature is not required for use,” said Bradley J. Monk, MD, FACS, FACOG, who was part of the development process as the clinical trial study was designed, enrolled, analyzed and published. “Although biomarkers are important in predicting clinical activity, the efficacy of ZEJULA is so broad that a companion diagnostic is not mandatory. However, germline BRCA testing is still important in identifying families at risk for hereditary breast and ovarian cancer.” Dr. Monk is an oncologist with Arizona Oncology, an affiliate of The US Oncology Network and US Oncology Research.
McKesson was also selected by TESARO as one of ZEJULA’s distributors of choice for in-office dispensing practices. Additionally, Biologics by McKesson, an independent specialty pharmacy with more than 24 years of experience connecting patients to life-changing medications in oncology and other complex therapeutic areas, was selected to be a specialty pharmacy provider in the ZEJULA distribution network. Eligible Onmark and Unity GPO members also have access to contract pricing.
In-office dispensing practices looking to access ZEJULA can place orders via McKesson’s Customer Center or by contacting their Customer Care team at 800.482.6700. Physicians may submit prescriptions to Biologics via phone (800.850.4306), fax (800.823.4506) or eScribe. For electronic prescribing systems, physicians may search for Biologics within their EMR system.
ZEJULA Important Safety Information
ZEJULA (niraparib) is a poly(ADP-ribose) polymerase (PARP) inhibitor indicated for the maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a complete or partial response to platinum-based chemotherapy. Myelodysplastic Syndrome/Acute Myeloid Leukemia (MDS/AML), including some fatal cases, was reported in patients treated with ZEJULA. Discontinue ZEJULA if MDS/AML is confirmed. Hematologic adverse reactions (thrombocytopenia, anemia and neutropenia), as well as cardiovascular effects (hypertension and hypertensive crisis) have been reported in patients treated with ZEJULA. Monitor complete blood counts to detect hematologic adverse reactions, as well as to detect cardiovascular disorders, during treatment. ZEJULA can cause fetal harm and females of reproductive potential should use effective contraception. Please see full prescribing information, including additional important safety information, available at http://www.zejula.com/.
About US Oncology Research
US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves 60 research sites and approximately 170 locations managing about 300 active trials at any given time. Physicians in the research network have enrolled more than 66,000 patients in over 1,500 trials since inception in 1992 and have played a role in nearly 70 FDA-approved cancer therapies, approximately one-third cancer therapies approved by the FDA to date. For more information visit https://www.usoncology.com/oncologists/us-oncology-research. To find clinical trials available through US Oncology Research visit http://trialfinder.usoncology.com
About Biologics by McKesson
Biologics by McKesson is an independent specialty pharmacy with over 24 years of experience connecting patients to life-changing medications in oncology and other complex therapeutic areas. Built on the foundation of deep clinical expertise and a high-touch approach to patient care, Biologics by McKesson delivers seamless access and personalized engagement, so patients get the most out of the care they receive. As part of McKesson Life Sciences, a business within McKesson Corporation, Biologics by McKesson harnesses unparalleled reach and connectivity across the healthcare system to connect the dots between payers, providers and biopharma, so together, they can deliver better care and outcomes for every patient.
About McKesson
McKesson Corporation, currently ranked 6th on the FORTUNE 500, is a global leader in healthcare supply chain management solutions, retail pharmacy, community oncology and specialty care, and healthcare information technology. McKesson partners with pharmaceutical manufacturers, providers, pharmacies, governments and other organizations in healthcare to help provide the right medicines, medical products and healthcare services to the right patients at the right time, safely and cost-effectively. United by our ICARE shared principles, our employees work every day to innovate and deliver opportunities that make our customers and partners more successful — all for the better health of patients. McKesson has been named the “Most Admired Company” in the healthcare wholesaler category by FORTUNE, a “Best Place to Work” by the Human Rights Campaign Foundation, and a top military-friendly company by Military Friendly. For more information, visit www.mckesson.com.
Media Contact
Claire Crye
281-825-9927
claire.crye@mckesson.com
###
Presentations will focus on innovative solutions for community oncology success and cancer care delivery in today’s challenging environment
The Woodlands, Texas (April 27, 2017) — Thought leaders from McKesson and The US Oncology Network (“The Network”) will play an important role in the upcoming 2017 Community Oncology Conference, the most comprehensive gathering in the country for independent oncology practices. The conference provides a wide range of networking and learning opportunities covering clinical, business and advocacy issues for community oncologists, providing the insight and knowledge they need to drive innovation and success in today’s evolving environment. Experts from McKesson and The Network will share their extensive knowledge of community oncology and the changing healthcare landscape by focusing on timely topics such as value-based care transformation, legislative issues impacting cancer care, and community oncology’s role in the Cancer Moonshot. Hosted by the Community Oncology Alliance (COA), the conference will be held April 27-28 in National Harbor, Maryland.
“While these are challenging times for all healthcare providers, as community oncologists we can find strength, unity and shared success through our common belief that patients are best served when they have the option to receive quality care in a community-based practice close to their home and support system,” said Debra Patt, MD, MPH, MBA, co-chair of the event and vice president of Public Policy and Academic Affairs for Texas Oncology, an affiliate of The US Oncology Network. “The Community Oncology Conference, like many similar meetings held by The Network, gives us the opportunity to share our successes and challenges and gather new ideas and solutions that can help all stakeholders in the cancer community thrive and succeed together.”
Conference attendees will learn about the latest treatment advances, the progress community oncology practices are making in implementing new payment reform models, as well as the role their work plays in fueling former Vice President Biden’s highly-touted Cancer Moonshot. As recognized leaders and innovators in community oncology, McKesson and The US Oncology Network will have a strong presence at the conference with stimulating presentations and panel participation from a variety of key thought leaders within the organizations, including:
-
Ben Jones, vice president of Government Relations & Public Policy, McKesson and The US Oncology Network. Jones will join the panel discussion, “Washington, DC Update: The Latest Legislative Issues Impacting Cancer Care.”
-
Joyce O’Shaughnessy, MD, medical oncologist with Texas Oncology and co-chair of Breast Cancer Research for The US Oncology Network. Dr. O’Shaughnessy will present on “Novel Therapies in Breast Cancer: HER2 Amplified, ER+, TNBC Disease.”
-
Debra Patt, MD, MPH, MBA, vice president of Public Policy and Academic Affairs with Texas Oncology, chair of The US Oncology Network Pathways Taskforce for Breast Cancer, editor in chief of ASCO’s JCO Clinical Cancer Informatics, and co-chair of the conference. Dr. Patt will serve as moderator for the panel discussion, “How Community Oncology is Fueling the Cancer Moonshot.”
-
R. Steven Paulson, MD, medical oncologist and president of Texas Oncology. Dr. Paulson will participate in the panel discussion focusing on the Cancer Moonshot.
-
Lalan Wilfong, MD, medical director of Quality Programs for Texas Oncology and physician champion of Value-Based Care for McKesson. Dr. Wilfong will speak on “Quantifying Physician Performance: Defining the Value Equation.” He is also participating in a panel discussion about practice benchmark reports and use of data.
Additionally, McKesson Specialty Health and The US Oncology Network are hosting an exclusive VIP reception at the conference featuring Ben Jones, McKesson’s expert in government relations and public policy. Jones will offer a quick update on the latest developments from Capitol Hill to help providers understand what they can expect over the next four years.
“The cancer community must remain vigilant and united in its efforts to address policies that impact the sustainability of community oncology,” said Jones. “Cancer is nonpartisan, so regardless of ideological perspective, we must continue to proactively engage on the issues that impact community oncology providers across the country. The strong commitment of McKesson and The US Oncology Network, as well as the strength we gain through our provider relationships and expertise, position us well to demonstrate our value and safeguard against changes that impact the welfare of cancer patients and the future of community oncology.”
For more information about the 2017 Community Oncology Conference or to register for the event, please visit the conference website at http://www.coaconference.org/.
About The US Oncology Network
Every day, The US Oncology Network (“The Network”) helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About McKesson
McKesson Corporation, currently ranked 6th on the FORTUNE 500, is a global leader in healthcare supply chain management solutions, retail pharmacy, community oncology and specialty care, and healthcare information technology. McKesson partners with pharmaceutical manufacturers, providers, pharmacies, governments and other organizations in healthcare to help provide the right medicines, medical products and healthcare services to the right patients at the right time, safely and cost-effectively. United by our ICARE shared principles, our employees work every day to innovate and deliver opportunities that make our customers and partners more successful — all for the better health of patients. McKesson has been named the “Most Admired Company” in the healthcare wholesaler category by FORTUNE, a “Best Place to Work” by the Human Rights Campaign Foundation, and a top military-friendly company by Military Friendly. For more information, visit www.mckesson.com.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@mckesson.com
# # #