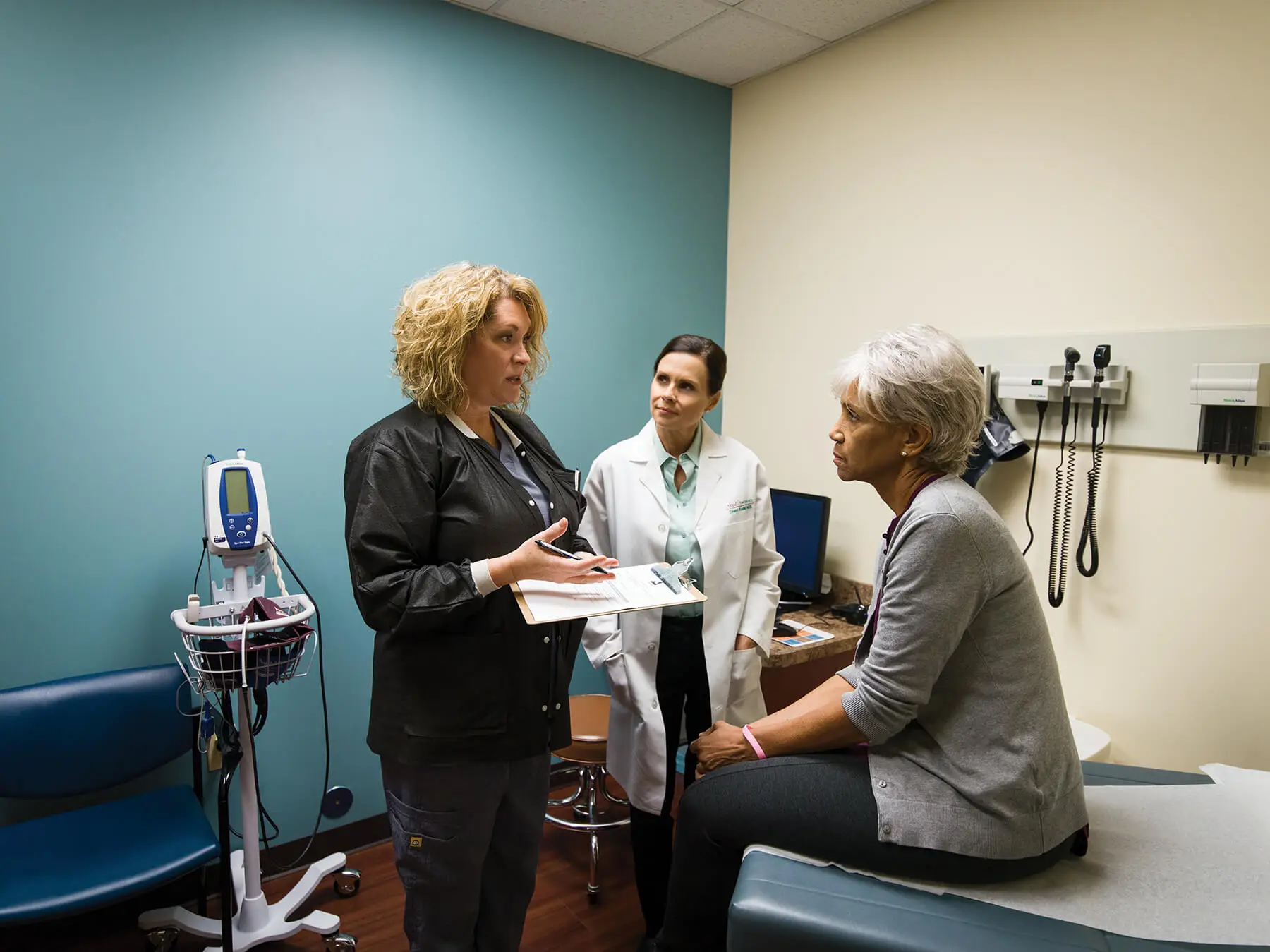
Program reaches recruitment milestone while bringing novel treatments to community cancer centers
The Woodlands, Texas (April 25, 2017) — US Oncology Research, one of the largest community-based oncology research programs in the United States, enrolled more than 2,900 subjects into clinical trials during the fiscal year ended March 31, 2017. The participation of these patients in leading-edge oncology trials for a wide range of cancer types contributed to eight cancer therapies receiving FDA approvals, making promising new treatments available to cancer patients across the nation. Contributing to so many FDA approvals in one fiscal year is unprecedented for US Oncology Research and demonstrates the organization’s strong ongoing commitment to play a vital role in understanding cancer and identifying effective therapies. In total, US Oncology Research has enrolled more than 66,000 patients into clinical trials since its inception in 1992.
“Accruing such a large number of patients in these novel clinical trials is a significant achievement for US Oncology Research, as finding appropriate patients, enrolling them into trials, and managing their complex care is very challenging,” said Michael Seiden, MD, PhD, chief medical officer, US Oncology Research. “These groundbreaking studies lay the foundation for the advancement of cancer care and are crucial to finding new and more effective ways to treat cancer. I am extremely proud of the world-class work done by our research team. Due to their dedication and commitment, patients in communities across the country now have access to some of the most encouraging therapies we have seen in years.”
According to the Cancer Research Institute, only three to six percent of cancer patients who are eligible for clinical trials take part in a study due to a variety of reasons, such as lack of awareness of the trial, difficulty with insurance, misinformation about the care provided during the study, or other concerns. US Oncology Research overcomes these recruitment barriers by employing a proven strategy that utilizes comprehensive models for the large-scale deployment of trials across select cities through affiliated community-based cancer centers.
One recruitment model the organization successfully uses is its Selected Trials for Accelerated Rollout (STAR) method, which contributed greatly to reaching last fiscal year’s 2,900 patient accrual milestone. This program opens and activates clinical trials quickly across all cancer centers affiliated with US Oncology Research. When a potential STAR trial patient is identified at a facility, the practice is trained in the details of the trial, and the study is opened within a two-week timeframe at the location where the patient will be treated.
Another factor that contributed to last year’s record accrual number was the organization’s ability to provide access to a vast number of immuno-oncology (I-O) trials, which comprise approximately one-third of the studies offered through US Oncology Research. Immunotherapy uses the body’s own immune system to help fight cancer. Today’s new I-O therapies have shown high efficacy rates, great tolerability and manageable toxicities. Additionally, some I-O trials have demonstrated durable responses even when therapy stops, suggesting the immune system may still be fighting the cancer even after treatment concludes. These dramatic trial results are generating significant excitement in the general media as well as the cancer community, and consequently, cutting-edge I-O trials―especially those in a community setting―are highly sought after by many patients.
The impact of the work done by US Oncology Research is far reaching, affecting many aspects of cancer care. Drug manufacturers rely on the organization to enroll patients in clinical trials to help bring new therapies to market. Promising new treatments can only become broadly available when safety and efficacy are demonstrated in clinical trials and presented as evidence for FDA approval. These approvals rely upon adequate patient participation in trials. Additionally, US Oncology Research-affiliated practices were able to offer 2,900 patients early access to exciting new therapies. Trial participation also helped these practices gain familiarity and achieve a comfort level with these investigational drugs prior to FDA approval, making post-approval prescribing easier.
“Clinical trials play a vital role in our commitment to provide the world’s most advanced cancer treatments to patients,” said David Smith, MD, director of Research for Compass Oncology and associate chair of the US Oncology Research Lung and Advanced Therapeutics Committees. “Patients being treated at a US Oncology Research-affiliated practice have convenient access to novel, investigational therapies in state-of-the-art cancer centers in communities close to home with their own oncologist directing their care. This supports an optimal outcome while providing a better patient and family experience.”
US Oncology Research recently released a video that highlights the contributions made to the advancement of cancer care.
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves 60 research sites and approximately 170 locations managing about 300 active trials at any given time. Physicians in the research network have enrolled more than 66,000 patients in over 1,500 trials since inception in 1992 and have played a role in nearly 70 FDA-approved cancer therapies, approximately one-third of all cancer therapies approved by the FDA to date. For more information visit https://www.usoncology.com/physicians/clinical-trials.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@usoncology.com
# # #
Practice with largest infusion treatment center in Delaware gains access to world-class treatments and enhances its cutting-edge research program
Newark, Del. (March 1, 2017) – Medical Oncology Hematology Consultants (“MOHC”), the premier provider of advanced treatments for cancer and blood disorder patients in Delaware for more than 30 years, has joined The US Oncology Network (“The Network”), the largest network of its kind comprised of community-based oncology practices dedicated to advancing high-quality, evidence-based cancer care. The Network is supported by McKesson Specialty Health, an industry leader in solutions that empower the community patient care delivery system to advance the science, technology and quality of care. By joining The Network, MOHC enhances its ability to provide patients high-quality care as well as access to some of the most advanced oncology treatments available today, all delivered in convenient neighborhood locations close to the patient’s home.
“Our goal is to provide the best care available to our patients by utilizing technology on the cutting edge, but tempered with clinical judgement and compassion,” said David Biggs, MD, medical oncologist/hematologist and managing partner, MOHC. “We believe that joining The US Oncology Network will strengthen our ability to achieve this goal by helping us to retain our independence and strengthen our operational capabilities. The comprehensive resources and guidance from The Network will enhance our ability to work closely with our patients and their family physicians to provide high-quality medical care and personal attention.”
The US Oncology Network includes more than 1,400 highly-credentialed medical oncology, hematology, radiation oncology, gynecologic oncology, urology, oncology surgery and other specialists. The addition of MOHC extends the reach of The Network, broadening and strengthening the capabilities and knowledge-base of the organization throughout the region. The Network’s collaboration with McKesson Specialty Health provides strong and comprehensive practice management capabilities and value-based care expertise critical for community practices navigating today’s challenging healthcare landscape.
MOHC has a well-established clinical research program and is one of ten community oncology practices to have been selected for an American Society of Clinical Oncology (ASCO) Clinical Trials Participation Award. By uniting with The Network, the practice will have the ability to greatly increase the number and variety of novel clinical trials it can offer patients through US Oncology Research, a leader in bringing innovative therapies to patients in local communities across the nation. The organization manages approximately 300 active trials at any given time and has played a role in more than 60 FDA-approved cancer therapies, about one-third of all those approved by the FDA to date.
“Medical Oncology Hematology Consultants is an outstanding example of how independent community oncology practices can provide high-quality patient care and compete successfully in the rapidly evolving healthcare environment,” said Kirk Kaminsky, president of The US Oncology Network and Practice Management for McKesson Specialty Health. “The US Oncology Network is committed to providing practices like MOHC with innovative and comprehensive practice management solutions, allowing providers to overcome business challenges and ensure long-term practice health so the physicians can focus on their top priority – helping their patients fight cancer.”
About Medical Oncology Hematology Consultants
Medical Oncology Hematology Consultants (“MOHC”) specializes in the diagnosis and treatment of cancer, blood disorders, thrombosis and related genetic disorders. MOHC physicians, nurses, practitioners and office personnel work closely with patients and their family doctors to provide them with the best in medical care and personal attention. Medical Oncology Hematology Consultants is now located on the campus of the Christiana Hospital in the Helen F. Graham Cancer Center West. For more information, visit www.cbg.org.
About The US Oncology Network
Every day, The US Oncology Network (“The Network”) helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@mckesson.com
Number One Rankings in Key Performance Areas Reinforces McKesson Specialty Health’s Leading Position in the Oncology and Hematology Market
The Woodlands, Texas (Feb. 28, 2017) — For the sixth year in a row, iKnowMedSM electronic health record (EHR) has been named the top-ranked EHR platform for oncologists and hematologists by Black BookTM Research, an unbiased industry-leading source for polling, surveys and market research. iKnowMed was recognized for its superior focus on meeting the unique needs of community-based oncology practices.
Implemented in nearly 650 sites of care nationwide and used by 1,700 providers, iKnowMed was the top-ranked oncology EHR across all practice sizes and delivery sites. The EHR platform received number one rankings in nine key performance areas – the most in this year’s report – including support and customer care, client relationships and cultural fit, reliability, best of breed technology and process improvement, and strategic alignment with client goals.
“Being the top-ranked oncology and hematology EHR for the sixth consecutive year is particularly rewarding because this recognition is based on independent customer satisfaction surveys and reflects iKnowMed’s ability to consistently meet and exceed the needs of oncology practices in an ever-changing healthcare environment,” said Dan Lodder, vice president and general manager of Technology Solutions for McKesson Specialty Health. “iKnowMed is a powerful EHR designed to help oncology practices operate more efficiently, boost financial performance and enhance the quality of patient care, all critical components for successfully transitioning to value-based care programs such as the Oncology Care Model (OCM) and the Medicare Access and CHIP Reauthorization Act of 2015 (MACRA).”
iKnowMed Generation 2, developed in collaboration with oncologists in The US Oncology Network and supported by McKesson Specialty Health, was the first next generation EHR for oncology and hematology available. This innovative EHR platform seamlessly integrates with McKesson Specialty Health’s leading technology solutions to meet the unique needs of oncology practices and enhance value-based care delivery while strengthening core practice operations.
“Black Book Research conducts a rigorous process to review technology and rank them accordingly,” said Doug Brown, managing partner, Black Book Research. “We are responsible for providing a trusted resource for physicians and administrators so they can make informed decisions about which product best meets their needs. We congratulate iKnowMed for its continued high ranking as the top EHR for Oncology and Hematology practices. It is an honor to be identified as a vendor that repeatedly puts customers first.”
Integrated iKnowMed modules include Clear Value PlusSM–clinical quality and regimen support; My Care PlusSM–an oncology-focused patient portal engaging more than 371,000 enrolled patients; Lynx Mobile® inventory and purchasing management; TotalViewSM Generation 2 revenue cycle reporting, and Practice InsightsSM analytics. Together, these best-in-class solutions help practices safely deliver high-quality patient care, advancing a practice’s ability to provide an integrated care experience for both patients and their comprehensive team of caregivers while optimizing revenue.
“We are excited that iKnowMed was not only recognized with the highest overall ranking but also the highest ratings in many key performance areas that are important to our customers. This reinforces our commitment to provide oncology practices award-winning, next-generation EHR solutions along with industry-leading support and service from our extremely talented and dedicated team,” said Calvin Chock, vice president of Product Management and Engineering for McKesson Specialty Health. “This ensures practices can confidently invest in one of the most innovative and robust technology platforms in oncology which drives efficiency and high-quality care.”
About McKesson Specialty Health
McKesson Specialty Health (“MSH”), a division of McKesson Corporation, works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care, passionately driven by the benefits it provides patients and the system as a whole. Through innovative provider, practice management, manufacturer and payer solutions, MSH focuses on improving the financial, operational and business health of our customers and partners so they may provide the best care to patients. At McKesson Specialty Health, we believe that we are all in this together. For more information, visit www.mckessonspecialtyhealth.com.
About The US Oncology Network
Every day, The US Oncology Network (“The Network”) helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About Black Book™
Black Book Market Research LLC, provides healthcare IT users, media, investors, analysts, quality minded vendors, and prospective software system buyers, pharmaceutical manufacturers, and other interested sectors of the clinical technology industry with comprehensive comparison data of the industry’s top respected and competitively performing technology vendors. The largest user opinion poll of its kind in healthcare IT, Black Book™ collects over 540,000 viewpoints on information technology and outsourced services vendor performance annually.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@mckesson.com
The Woodlands, Texas (February 2, 2017) — Representatives from McKesson and The US Oncology Network (The Network) will be presenting at the 10th Cancer Center Business Summit, which will be held Monday and Tuesday, February 6-7 at The Aria Resort in Las Vegas, Nevada. The Summit aims to showcase innovative organizations and how they are responding to changes occurring in the cancer care marketplace.
“Today’s community oncology practices face many decisions and challenges, from new reimbursement structures to care models and technologies,” said Randy Hyun, senior vice president and general manager, Provider Solutions, McKesson. “With these decisions come immense opportunities, and the Summit provides an important forum for discussion and knowledge-sharing. We are thrilled to have several panelists, representing multiple disciplines, participate in this year’s Summit. Our team will share their unique perspectives and insights on how organizations can remain independent in today’s consolidating landscape to best practices across a variety of important subjects, including clinical pathways and the use of big data.”
Marcus Neubauer, MD, vice president and medical director, Payer and Clinical Services, McKesson and The US Oncology Network, will participate in the Summit’s “Clinical Pathways: With What Result?” panel and discuss The Network’s experience with the Oncology Care Model (OCM) as it relates to clinical pathways. The OCM is a payment initiative introduced by the Centers for Medicare & Medicaid Services (CMS) to support the transition to value-based care. Dr. Neubauer will outline how practices can be successful under OCM and how adherence to clinical pathways will remain a cornerstone for value-based care. He will also provide real-world knowledge garnered from more than a decade of The Network’s experience with clinical pathways and, more recently, the OCM.
“The incorporation of clinical pathways is an important component for success in the OCM and other value-based care models,” said Dr. Neubauer. “The ability to transform and meet program requirements, provide enhanced services, and negotiate agreements with payers that support these aligned incentives all play critical roles in a practice’s performance and need to work in unison. We welcome the opportunity to share key learnings from our experience with value-based reimbursement models as we all strive to navigate today’s ever-changing cancer care landscape.”
Representatives from McKesson and members of The Network will participate in Summit panels discussing a broad spectrum of topics affecting the ecosystem of oncology practices, including value-based care and OCM, use of health technology and data, and important considerations for sustainable business models in today’s consolidating market. In addition, specialists in the areas of Merit-based Incentive Payment System (MIPS) and the Medicare Access and CHIP Reauthorization Act of 2015 (MACRA) will be on hand to discuss the latest trends and demands on practice leaders in the area of reimbursement.
Presenters from McKesson and The US Oncology Network include:
- Randy Hyun, senior vice president and general manager, Provider Solutions, McKesson will participate in the Summit’s “Remaining Independent: Achieving Scale through Practice Mergers & Collaborative Networks” panel discussion.
- Diana Verrilli, senior vice president, Payer and Practice Management Solutions, McKesson will join the discussion on “The Oncology Care Model: Evolving Best Practices.”
- Marcus Neubauer, MD, vice president and medical director, Payer and Clinical Services, McKesson and The US Oncology Network will participate in the “Clinical Pathways: With What Result?” panel.
- Debra Patt, MD, MPH, MBA, vice president of Public Policy and Academic Affairs at Texas Oncology and chair of The US Oncology Network Pathways Taskforce for Breast Cancer and editor in chief of ASCO’s JCO Clinical Cancer Informatics, is slated to participate in the Summit’s “Big Data Platforms to Support Community Oncology” panel.
- J. Russell Hoverman, MD, PhD, vice president of Quality Programs at Texas Oncology and medical director of Managed Care for McKesson and The US Oncology Network will share his perspective on palliative care solutions during a session of the same name.
The above panelists and other subject matter experts will be available in the McKesson exhibit space during the Summit. Additional information about the programs featured during the Summit can also be found at www.mckesson.com and www.usoncology.com.
About The US Oncology Network
Every day, The US Oncology Network helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About McKesson
McKesson Corporation, currently ranked 6th on the FORTUNE 500, is a global leader in healthcare supply chain management solutions, retail pharmacy, community oncology and specialty care, and healthcare information technology. McKesson partners with pharmaceutical manufacturers, providers, pharmacies, governments and other organizations in healthcare to help provide the right medicines, medical products and healthcare services to the right patients at the right time, safely and cost-effectively. United by our ICARE shared principles, our employees work every day to innovate and deliver opportunities that make our customers and partners more successful — all for the better health of patients. McKesson has been named the “Most Admired Company” in the healthcare wholesaler category by FORTUNE, a “Best Place to Work” by the Human Rights Campaign Foundation, and a top military-friendly company by Military Friendly. For more information, visit www.mckesson.com.
Media Contact
Claire Crye
McKesson
281-825-9927
Claire.Crye@mckesson.com
Edie DeVine
GCI Health on behalf of McKesson
209-814-9564
Edie.DeVine@gcihealth.com
Affiliation with the largest network of its kind strengthens Southern Cancer Center’s ability to remain independent, providing access to vast resources to enhance care
Mobile, Ala. (February 1, 2017) – Southern Cancer Center, a physician-owned multi-site medical oncology practice serving the Alabama Gulf Coast region, has joined The US Oncology Network (“The Network”). The Network provides strong and comprehensive practice management capabilities and value-based care expertise that are critical for independent practices such as Southern Cancer Center navigating today’s challenging healthcare landscape. The Network is supported by McKesson Specialty Health, an industry leader in solutions that empower the community patient care delivery system to advance the science, technology and quality of care. Southern Cancer Center and The US Oncology Network share a vision of keeping independent physician groups strong and viable, while aiming to improve patient health.
“Our top priority was, is and always will be the health of our patients,” said Michael W. Meshad, M.D., president and founder of Southern Cancer Center. “That’s why joining forces with The US Oncology Network was an easy choice and the right fit for our practice. I, as well as my fellow physicians and care team, can focus on what’s most important, our patients, all while knowing that the health and viability of our practice is being cared for by The Network. Additionally, because of this affiliation, our practice can remain independent and physician-owned, caring for patients in the community in which they live.”
As part of The US Oncology Network, Southern Cancer Center will have access to expertise and resources from The Network typically only available in large academic medical centers. The practice will have access to state-of-the-art technology and programs to support oncology practices as they transition to value-based care delivery models, including iKnowMedSM Generation 2 electronic health record and Clear Value PlusSM chemotherapy regimen support tool. Additionally, physicians will be able to collaborate on difficult cases with their colleagues across America through eTumor Boards and The Oncology Portal, tapping into the collective knowledge and experience of more than 1,400 physicians.
Southern Cancer Center will participate in clinical trials through US Oncology Research, a leader in bringing innovative therapies to patients in local communities across the nation. The organization manages approximately 300 active trials at any given time and has played a role in more than 60 FDA-approved cancer therapies, about one-third of all those approved by the FDA to date.
The Network also benefits from the collaboration. The addition of Southern Cancer Center’s eight highly-credentialed and specialized medical oncology physicians extends the reach of The Network, broadening and strengthening the capabilities and knowledge-base of the organization throughout the Gulf Coast Region of Alabama.
“The US Oncology Network is committed to supporting independent community oncology practices to remain viable and thrive in a complex and evolving healthcare environment,” said Kirk Kaminsky, president of The US Oncology Network and Practice Management for McKesson Specialty Health. “Our innovative and comprehensive practice management solutions allow providers to overcome business challenges and ensure long-term practice health so the physicians can focus on their top priority – helping their patients fight cancer. We are excited for Southern Cancer Center to join the Network and look forward to our work together toward our common goal of improving healthcare.”
About Southern Cancer Center
Southern Cancer Center is a multi-site medical oncology practice founded by established Mobile-area physicians. Southern opened in 2007 with the desire to offer a patient centered healthcare approach. Current offerings include Hematology/Oncology, in-house laboratory, second opinions, in-house specialty retail pharmacy, flow cytometry, and access to all local hospitals. The practice serves patients from five convenient locations throughout the area, including three sites in Mobile at Providence Hospital, Springhill Medical Center and Mobile Infirmary as well as Daphne and Foley in Baldwin County. For more information about Southern Cancer Center, visit www.southerncancercenter.com. Appointments for all services can be scheduled by calling 1-800-625-6896 or 251-625-6896.
About The US Oncology Network
Every day, The US Oncology Network helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
Media Contact
Claire Crye
The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
Hetal Bhatia
McKesson Specilaty Health
415-760-6957
Hetal.Bhatia@mckesson.com
The Woodlands, Texas (January 26, 2017) – The US Oncology Network (“The Network”) announced today, as part of McKesson Specialty Health’s ongoing integration with Vantage Oncology, that 50 centers in 13 states and more than 350 affiliated physicians have joined The Network.
The US Oncology Network provides effective and comprehensive practice management capabilities and value-based care expertise critical for navigating today’s challenging healthcare landscape. The Network is supported by McKesson Specialty Health, an industry leader that works together with stakeholders across the healthcare delivery system to preserve and strengthen specialty care.
“Complexity for independent, community-based providers continues to increase as the healthcare landscape evolves. Our innovative and comprehensive practice management solutions coupled with the power of 1,400 like-minded physicians ease provider challenges and ensure long-term practice health,” said Kirk Kaminsky, president of The US Oncology Network and Practice Management for McKesson Specialty Health. “As we integrate Vantage Oncology practices into The US Oncology Network, we will be better positioned than ever to empower providers in our physician-led organization to focus on advancing patient care.”
With Vantage Oncology and its practices now part of The US Oncology Network, The Network includes more than 1,400 highly-credentialed medical oncology, hematology, radiation oncology, gynecologic oncology, urology, oncology surgery and other specialists. The Network and its affiliated physicians treat more than 800,000 patients annually at over 400 sites of care in more than 25 states. The addition of these centers extends the reach of The Network, broadening and strengthening the capabilities and knowledge-base of the organization throughout the country.
“Our centers’ mission remains to preserve the patient-doctor relationship and provide access to a high-quality healthcare experience,” said Charles Smith, Ph.D., chief operating officer, Vantage Oncology. “By becoming part of The US Oncology Network, we continue to pursue this mission with the expanded scale, resources, infrastructure, and expertise we require to remain strong and independent.”
Centers joining The US Oncology Network include:
- Gulf Coast Cancer Centers – Brewton, AL
- Gulf Coast Cancer Centers – Daphne, AL
- Gulf Coast Cancer Centers – Foley, AL
- Northern Arizona Tumor Institute – Prescott, AZ
- Northern Arizona Tumor Institute – Prescott Valley, AZ
- Imperial Valley Cancer Center – El Centro, CA
- Glendale Radiation Therapy Center – Glendale, CA
- Los Angeles Radiation Oncology Center – Los Angeles, CA
- West Hills Radiation Therapy Center – West Hills, CA
- Redhawk Radiation Therapy Center – Temecula, CA
- Riverside Radiation Therapy Center – Riverside, CA
- St. Bernardine Radiation Therapy Center – San Bernardino, CA
- Wildomar Radiation Therapy Center – Wildomar, CA
- Center for Radiation Therapy of Beverly Hills – Beverly Hills, CA
- Holy Cross Radiation Therapy Center – Mission Hills, CA
- Santa Clarita Radiation Therapy Center – Valencia, CA
- Sherman Oaks Radiation Therapy Center – Sherman Oaks, CA
- Radiotherapy Clinics of Georgia – Conyers, GA
- Radiotherapy Clinics of Georgia – Covington, GA
- Radiotherapy Clinics of Georgia – Decatur, GA
- Cancer Center at Blairsville – Blairsville, GA
- Gwinnett Radiation Oncology – Lawrenceville, GA
- Radiotheraphy Clinics of Georgia – Lawrenceville, GA
- Radiotheraphy Clinics of Georgia – Snellville, GA
- Cancer Center at Lawrenceville – Lawrenceville, GA
- InterCommunity Cancer Center – Lady Lake, FL
- Southeast Regional Prostate Cancer Treatment Center – Daytona Beach, FL
- Cancer Care Centers of Brevard – Melbourne, FL
- Cancer Care Centers of Brevard: Wickham – Melbourne, FL
- Cancer Care Centers of Brevard – Sebastian, FL
- Cancer Care Centers of Brevard – Merritt Island, FL
- Integrated Cancer Care – Indianapolis, IN
- Integrated Cancer Care – Greenwood, IN
- Evansville Cancer Center – Evansville, IN
- Deaconess Chancellor Center for Oncology – Newburgh, IN
- Radiotherapy Centers of Kentuckiana – Jeffersonville, IN
- Radiotherapy Centers of Kentuckiana – Louisville, KY
- South Suburban Cancer Center – Hazel Crest, IL
- Southland Oncology – Mokena, IL
- Valley Regional Cancer Center – Peru, IL
- Streator Radiation Oncology – Streator, IL
- Signature Healthcare Radiation Therapy Center – Brockton, MA
- New York Treatment Center – Brooklyn, NY
- New York Treatment Center – Flushing, NY
- Columbus Prostate Cancer Center – Gahanna, OH
- Comprehensive Cancer Care – Exton, PA
- Commonwealth Health Cancer Center – Wilkes-Barre, PA
- NorthMain Radiation Oncology – Providence, RI
About The US Oncology Network
Every day, The US Oncology Network helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson Specialty Health. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
Media Contact
Claire Crye
The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
Dr. Debra Patt shared expertise on the event’s Support Services and Survivorship Panel
The Woodlands, Texas (January 11, 2017) — Representatives from McKesson and The US Oncology Network (The Network) were invited to participate in a community oncology event as part of the White House “Making Health Care Better” Series, which highlights progress made in improving the health system over the past eight years. The President’s Health Policy Team collaborated with the Cancer Moonshot program to host the event and put a spotlight on the role and importance of community oncology in cancer care. The event was held on Wednesday, January 11 from 8am to 12pm ET at the White House and was only open to invited attendees.
Debra Patt, MD, MPH, MBA, vice president of Public Policy and Academic Affairs at Texas Oncology and chair of The US Oncology Network Pathways Taskforce for Breast Cancer, participated in the event’s Support Services and Survivorship Panel.
“This was an exciting opportunity to collaborate with others who are passionate about improving healthcare as well as showcase the importance of community cancer care,” said Dr. Patt. “Most cancer care occurs in the community setting, and many patients can receive state-of-the-art care in their home towns, including access to clinical trials. This allows patients to be treated in and supported by their communities. Additionally, research shows that outpatient services cost significantly more when provided in the outpatient hospital setting without any meaningful improvement in quality of care or health outcomes. This is one of many reasons healthcare stakeholders must continue events like this and learn about progress and innovation in community cancer care.”
The event focused on improving innovation and access to quality cancer care in the community setting and featured panels to explore the unique aspects of community oncology, including cancer disparities, survivorship and support services, advancing clinical trials and innovative models of care delivery in the community setting, and the future of healthcare for people affected by cancer.
Representatives from McKesson and The US Oncology Network invited to attend the event include:
- Ben Jones, vice president, Government Relations & Public Policy, McKesson
- Diana Verrilli, senior vice president, Payer and Practice Management Solutions, McKesson
- Nicholas J. Robert, MD, medical director, Health Economic Outcomes Research and Health Informatics, McKesson and The US Oncology Network
- Debra Patt, MD, MPH, MBA, vice president of Public Policy and Academic Affairs at Texas Oncology and chair of The US Oncology Network Pathways Taskforce for Breast Cancer
- R. Steven Paulson, MD, president of Texas Oncology, a practice in The US Oncology Network
- Alex Spira, MD, PhD, FACP, director of the Virginia Cancer Specialists Research Institute and chair of The US Oncology Network Pathways Taskforce for Lung Cancer
- Sandy Smith, senior director, Site Services, US Oncology Research and McKesson
To continue the conversation around community oncology, immediately following the event McKesson and The US Oncology Network are hosting a Hill briefing in collaboration with the Association of Community Cancer Centers (ACCC), American Cancer Society-Cancer Action Network, Cancer Support Community, Community Oncology Alliance (COA) and Sarah Cannon. Key members of Congress plan to attend the briefing including Congresswoman Diana DeGette (D-CO) and Congressman Fred Upton (R-MI).
“McKesson is committed to working with all stakeholders across the healthcare system to strengthen care as we are all in this together,” said Nick Loporcaro, president, McKesson. “Being invited to the White House ‘Making Health Care Better’ Series is a testament to the leading role McKesson and The US Oncology Network are playing in the industry. Our people and their expertise and dedication help drive the movement of improving and sustaining specialty care in the community setting.”
About The US Oncology Network
Every day, The US Oncology Network helps more than 1,400 independent physicians deliver value-based, integrated care for patients — close to home. Through The Network, these independent doctors come together to form a community of shared expertise and resources dedicated to advancing local cancer care and to delivering better patient outcomes. The Network provides doctors with access to coordinated resources, best business practices, and the experience, infrastructure and support of McKesson. This collaboration allows the doctors in The Network to focus on the health of their patients, while McKesson focuses on the health of their practices. Together, The Network and its affiliated physicians are committed to the success of independent practices, everywhere. For more information, visit www.usoncology.com.
About McKesson
McKesson Corporation, currently ranked 6th on the FORTUNE 500, is a global leader in healthcare supply chain management solutions, retail pharmacy, community oncology and specialty care, and healthcare information technology. McKesson partners with pharmaceutical manufacturers, providers, pharmacies, governments and other organizations in healthcare to help provide the right medicines, medical products and healthcare services to the right patients at the right time, safely and cost-effectively. United by our ICARE shared principles, our employees work every day to innovate and deliver opportunities that make our customers and partners more successful — all for the better health of patients. McKesson has been named the “Most Admired Company” in the healthcare wholesaler category by FORTUNE, a “Best Place to Work” by the Human Rights Campaign Foundation, and a top military-friendly company by Military Friendly. For more information, visit www.mckesson.com.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@usoncology.com
When Fully Staffed, the McKesson Indiana Regional Billing Office will Employ More than 100
EVANSVILLE, Indiana (December 13, 2016) — Evansville Mayor Lloyd Winnecke and McKesson announced that McKesson will expand its Revenue Cycle Services operations with the opening of a new office in Evansville, Indiana in early 2017. When fully staffed, the new McKesson Indiana Regional Billing Office will employ more than 100.
“I am excited that McKesson has chosen Evansville to locate this new office and create additional jobs for the City and the region,” said Mayor Winnecke. “This reinforces our focus and efforts of fostering job creation and business growth, and I expect McKesson’s industry leadership will serve as an invaluable contribution to the Indiana economy.”
McKesson’s reimbursement and revenue cycle management group provides comprehensive practice management solutions to help medical groups and physician offices improve their financial performance and efficiency. The Evansville office will serve practices affiliated with The US Oncology Network, which is supported by McKesson, an industry leader in solutions that empower the community patient care delivery system to advance the science, technology and quality of care.
“McKesson is committed to helping our customers improve the efficiency, effectiveness and productivity of their reimbursement and revenue-cycle management functions, leaving more time to care for patients,” said Greg Tenbarge, VP, Revenue Cycle Services, McKesson. “We have worked closely with Mayor Winnecke and the Southwestern Indiana Chamber of Commerce on this expansion, and we are very excited to tap into the highly-qualified local workforce and look forward to being an active part of the Evansville community.”
The 13,900-square foot facility will feature benefits and amenities including a convenient location, 401 SE 6th Street, Evansville, Indiana 47713. It’s designed to be a space that inspires engagement, creativity, collaboration and wellness for the employees based there. This new building is a great example of McKesson’s continued focus on investing in its workforce.
“With the building of the downtown Evansville medical and research campus to new investments being made in the Warrick Wellness Trail Medical District, the region’s life science and healthcare sectors continue to grow, creating not only employment opportunities, but a competitive footprint for Indiana’s Great Southwest,” said Greg Wathen, President & CEO of the Economic Development Coalition of Southwest Indiana.
About McKesson
McKesson Corporation, currently ranked 6th on the FORTUNE 500, is a global leader in healthcare supply chain management solutions, retail pharmacy, community oncology and specialty care, and healthcare information technology. McKesson partners with pharmaceutical manufacturers, providers, pharmacies, governments and other organizations in healthcare to help provide the right medicines, medical products and healthcare services to the right patients at the right time, safely and cost-effectively. United by our ICARE shared principles, our employees work every day to innovate and deliver opportunities that make our customers and partners more successful — all for the better health of patients. McKesson has been named the “Most Admired Company” in the healthcare wholesaler category by FORTUNE, a “Best Place to Work” by the Human Rights Campaign Foundation, and a top military-friendly company by Military Friendly. For more information, visit www.mckesson.com.
Media Contact
Claire Crye
281-825-9927
Claire.Crye@mckesson.com
15th Annual Science Forum promotes clinical research education, innovation and collaboration
The Woodlands, Texas (December 8, 2016) — US Oncology Research held its 15th Annual Science Forum earlier this fall. The annual meeting for all US Oncology Research affiliated investigators was held in Dallas and highlighted the importance of clinical trials as a treatment option for cancer patients.
“A clinical trial saved my life,” said Rhonda Jenkins, breast cancer survivor, clinical trial participant and patient of Texas Oncology, an affiliate of US Oncology Research.
Jenkins shared her story with the hundreds of clinical attendees, including investigators and research coordinators. “The best advice I can give to patients is to not turn down a clinical trial if one is available. I received a terminal diagnosis eleven years ago. Because of a trial, I have my life back. I no longer consider myself as having a terminal disease. Plus, participating in a trial allowed me to play a part in hopefully helping future patients fight this disease,” added Jenkins.
The Annual Science Forum allows research investigators to discuss and exchange thoughts on the latest scientific approaches and novel treatment strategies. Topics of discussion included: immunotherapy; the oncology care model; early phase trials; proton therapy; genomics; precision medicine; and much more.
“This Forum represents an important form of education for all of us in the cutting-edge, ongoing work in basic and clinical research against cancer,” said Daniel Von Hoff, MD, FACP, chief scientific officer, US Oncology Research. “It helps our team craft strategies for the best way to help our patients.”
One strategy used by US Oncology Research is its Selected Trials for Accelerated Rollout, or STAR, method. This program quickly opens clinical trials for difficult-to-find patients. When a potential STAR trial patient has been identified, the practice is trained and the study is opened within a two-week timeframe at the location where the patient is expected to be seen. Ultimately, STAR provides the latest in clinical research to the practice where the patient has been identified.
“We are truly delivering tomorrow’s treatments today,” said Michael Seiden, MD, chief medical officer, The US Oncology Network and McKesson Specialty Health. “The Annual Science Forum brings together the leadership of US Oncology Research as we both celebrate our accomplishments and look forward to building an even stronger culture of research across our national network of affiliated physicians and research staff.”
During the Forum, investigators convened to hear from world renowned speakers on topics and trends in oncology research. The purpose is to continue to be at the cutting edge of research and provide patients in the community setting access to the latest treatments. Roy Herbst, MD, PhD, chief of Medical Oncology and associate director for Translational Research at Yale Cancer Center, was a keynote speaker at the Forum and spoke about novel therapies for lung cancer, including targeted and immuno-therapies.
“Collaborating with research investigators across the country on the most recent trends in oncology treatment is extremely important,” said Dr. Herbst. “It is our responsibility as researchers and providers to constantly be in search for the latest and most effective treatment options to improve efficacy and decrease toxicity. It was an honor to be among such passionate and dedicated clinicians and researchers.”
“This year’s Annual Science Forum really put a charge into our passion for clinical research,” said David Cosgrove, MD, oncologist with Compass Oncology and associate chair of the US Oncology Research Gastrointestinal (GI) Committee. “We heard from impressive speakers involved in cutting-edge science, highlighted recent successes in our field and got great perspective regarding patient communication. The Forum highlighted some of the challenges we will face in clinical research in a community oncology setting in the years ahead, and also provided a robust framework for future success building upon the US Oncology Research model that leads the way in these endeavors. I am excited to be part of this group moving forward.”
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves approximately 60 research sites and nearly 160 locations managing about 300 active trials at any given time. Physicians in the research network have enrolled more than 64,000 patients in more than 1,500 trials since inception in 1992 and have played a role in more than 60 FDA-approved cancer therapies, nearly one-third of all cancer therapies approved by the FDA to date. For more information about US Oncology Research visit https://www.usoncology.com/oncologists/us-oncology-research. To find clinical trials available through US Oncology Research visit http://trialfinder.usoncology.com.
Media Contact
Claire Crye
The US Oncology Network
281-825-9927
claire.crye@usoncology.com
Collaboration strengthens both practices, enhancing patient care, research opportunities and access to services throughout Northern Florida
Ocala, Fla. (December 1, 2016) — Florida Cancer Affiliates (FCA), a premier provider of advanced cancer care services in Florida, announced that Blood & Cancer Center has joined its practice. Anju Vasudevan, M.D., and Cecil Robertson, M.D., oncologists and hematologists with Blood & Cancer Center, will unite with Florida Cancer Affiliate’s group of physicians to provide comprehensive state-of-the-art cancer care in convenient, patient-friendly community settings. FCA is an affiliate of The US Oncology Network (The Network), one of the nation’s largest networks of integrated community-based oncology practices.
“Our first priority is to provide high-quality care to our patients,” said Dr. Vasudevan, founding member of Blood & Cancer Center. “By uniting with Florida Cancer Affiliates and The US Oncology Network, we can continue building on our tradition of excellence by enhancing our patient care services as well as expanding our clinical trial program through US Oncology Research, one of the largest community-based oncology research programs in the United States.”
By joining FCA, Blood & Cancer Center physicians can collaborate with an expanded physician community of more than 1,000 through The US Oncology Network. As members of The Network, physicians and clinicians share expertise and best practices through committees, workshops, conferences and via The Oncology Portal, a secure on-line portal where physicians can share data and collaborate on complex cases. Blood & Cancer Center physicians will also be able to utilize Value Pathways powered by NCCNTM—evidence-based treatment guidelines developed by physicians and clinicians in The Network together with the National Comprehensive Cancer Network® (NCCN®).
Blood & Cancer Center also gains access to innovative technologies and services that will further support their operational efficiency. Additionally, The Network provides strong and comprehensive practice management capabilities and value-based care expertise that are critical for practices navigating today’s challenging healthcare landscape.
Drs. Vasudevan and Robertson have decades of experience in cancer care. Dr. Vasudevan completed her residency at Bergen Pines County Hospital in Paramus, New Jersey and was a fellow at the Roswell Park Cancer Institute and Buffalo General Hospital in Buffalo, New York. Dr. Robertson received his medical degree at State University of New York at Buffalo and completed a combined hematology and oncology fellowship at Queens Hospital Center.
“Drs. Vasudevan and Robertson are extremely well respected in the cancer care community and having them join our practice makes our team even stronger, enhancing our ability to serve the patient population with high-quality, compassionate care,” said Rama Balaraman, M.D., oncologist with FCA. “Our collaboration with these like-minded, highly-trained physicians who share our vision of providing leading-edge community-based care is a perfect fit.”
About Florida Cancer Affiliates
Florida Cancer Affiliates (FCA) is a community-based radiation oncology, medical oncology, and hematology practice with convenient locations throughout Florida, from Naples and Ft. Myers to Trinity, Hudson, Brooksville, The Villages, Ocala and Panama City. The integrated clinical team of FCA employs the latest technologies and drug therapies to ensure patients receive advanced care close to home. The comprehensive treatment centers include outpatient treatment and diagnostic facilities providing chemotherapy, hematology services, radiation therapy including stereotactic and PET/CT imaging, clinical research, pharmacy and laboratory, as well as financial counseling and patient support services.
The practice is committed to providing patients with advanced care. They offer therapies proven to be effective, along with advanced diagnostic and treatment technologies. They also provide access to new investigational drugs through clinical trials. By participating in clinical trials that test new drugs or various combinations of treatments, patients have the opportunity to receive new therapies not yet available outside these clinical trials. To learn more about Florida Cancer Affiliates, go to www.floridacancer.com.
Florida Cancer Affiliates is united in healing with The US Oncology Network, one of the nation’s largest networks of integrated community-based oncology practices dedicated to advancing high-quality, evidence-based cancer care. As an affiliate of The US Oncology Network, FCA is united with more than 1,000 physicians nationwide. Florida Cancer Affiliates participates in clinical trials through US Oncology Research, which has played a role in more than 60 FDA-approved cancer therapies, nearly one-third of all cancer therapies approved by the FDA to date. For more information, visit www.usoncology.com.
Claire Crye
The US Oncology Network
281-825-9927
Claire.Crye@usoncology.com
Sixteen presentations on clinical research conducted by top US Oncology Research affiliated investigators will take place at ASH 2016
The Woodlands, Texas (November 30, 2016) — More than 20 leading investigators affiliated with US Oncology Research participated in sixteen studies that will be presented at the 58th American Society of Hematology Annual Meeting and Exposition (ASH), a premier scientific event in malignant and non-malignant hematology, held December 3-6 in San Diego. Six oral and ten poster presentations co-authored by US Oncology Research affiliated investigators cover topics such as diffuse large B-cell lymphoma (DLBCL), multiple myeloma, small lymphocytic leukemia, and mantle cell lymphoma.
David Andorsky, MD, oncologist and hematologist with Rocky Mountain Cancer Centers, an affiliate of US Oncology Research, will be presenting results from a lymphoma study titled, “MAGNIFY: Phase IIIb Randomized Study of Lenalidomide Plus Rituximab (R2) Followed By Lenalidomide Vs. Rituximab Maintenance in Subjects with Relapsed/Refractory Follicular, Marginal Zone, or Mantle Cell Lymphoma.” The poster presentation will take place Saturday, December 3 from 5:30 to 7:30pm PT in Hall GH.
“This is an important study for patients being treated for indolent B-cell lymphoma,” said Dr. Andorsky. “R2 induction therapy shows favorable activity and a good safety profile for many patients. This is a preliminary report, and further follow up will be needed to demonstrate the efficacy and safety of this regimen.”
Jeff Sharman, MD, director of Research at Willamette Valley Cancer Institute and Research Center and medical director of Hematology Research for The US Oncology Network, will be presenting results from a mantle cell lymphoma study titled, “Results of a Phase 2 Trial Evaluating Efficacy and Safety of Entospletinib (GS-9973) in Patients with Mantle Cell Lymphoma.” The poster presentation will take place Sunday, December 4 from 6:00 to 8:00pm PT in Hall GH.
“Research for hematologic malignancies in the community setting is crucial for the advancement of cancer treatment,” said Dr. Sharman. “I’m excited about the results being presented this year and hope it will lead to additional treatment options for patients.”
Also among the findings co-authored by investigators affiliated with US Oncology Research is the ongoing Phase 2 study of Brentuximab Vedotim with Rchp as frontline therapy in patients with High-Intermediate/High-Risk DLBCL. Results of the trial, co-authored in part by 11 US Oncology Research affiliated physicians, will be featured as an oral presentation on Saturday, December 3 at 9:45am PT in Room 7AB.
“It’s outstanding to see the cutting-edge research conducted by physicians affiliated with US Oncology Research each and every year,” said Michael Seiden, MD, chief medical officer, The US Oncology Network and McKesson Specialty Health. “The patient is top priority for these physicians, which is evidenced by the quality and caliber of their work and the level of their dedication. Research is the cornerstone of advancing treatment, and the physicians presenting at this year’s ASH are leading the way.”
The full schedule of US Oncology Research affiliated data presentations, including location information, can be found here. For more information or to interview a trial investigator, contact Claire Crye at 281-825-9927 or Claire.Crye@usoncology.com or Kerry Sinclair at 323-602-1096 or Kerry.Sinclair@gcihealth.com.
About US Oncology Research
Supported by McKesson Specialty Health and The US Oncology Network, US Oncology Research draws from a network of experienced investigators and dedicated clinical staff who specialize in oncology clinical trials. US Oncology Research serves approximately 60 research sites and nearly 160 locations managing about 300 active trials at any given time. Physicians in the research network have enrolled more than 64,000 patients in more than 1,500 trials since inception in 1992 and have played a role in more than 60 FDA-approved cancer therapies, nearly one-third of all cancer therapies approved by the FDA to date. For more information visit www.usoncology.com/oncologists.
Media Contacts
Claire Crye
US Oncology Research
281-825-9927
Claire.Crye@usoncology.com
Kerry Sinclair
GCI Health on behalf of US Oncology Research
323-602-1096Kerry.Sinclair@gcihealth.com
Denver, Colo. (November 15, 2016) – Rocky Mountain Cancer Centers (RMCC), a practice in The US Oncology Network, is the first in the Rocky Mountain region, and one of only a handful of sites in the United States, to offer the newly approved Ga-68 DOTATATE PET/CT for patients with neuroendocrine tumors (NETs). The U.S. Food and Drug Administration (FDA) recently approved the test. Marketed under the name Netspot™, gallium 68 DOTATATE is a radioactive diagnostic agent used in positron emission tomography (PET) imaging. Because the gallium DOTATATE binds strongly to the receptors of neuroendocrine tumor cells, it effectively detects tumors and metastases, not seen on other standard scans, and reveals the locations of tumors.
The Ga-68 DOTATATE scan allows surgeons to pinpoint the precise location of tumors before beginning surgery. RMCC neuroendocrine tumor surgeon, and internationally recognized disease expert, Eric Liu, MD, FACS, was instrumental in research that led to the approval of the gallium diagnostic test. “The information is critical for me to do the best possible surgery for patients,” Dr. Liu noted.
Nearly 10,000 people in the United States are diagnosed with NETs each year. The tumors are slow-growing but very likely to spread. They primarily affect abdominal and respiratory organs, including the stomach, pancreas, intestines, and lungs. The condition historically has been difficult to diagnose, and the cancer is not well recognized or understood across the medical community.
“The key to effective treatment of a rare cancer like neuroendocrine is accurate diagnosis,” says Allen Cohn, MD, co-director of Medical Services of The Neuroendocrine Institute at Rocky Mountain Cancer Centers. “The level of detail provided by the Ga-68 DOTATATE scan allows us to more accurately stage the disease. We can then provide our patients with a treatment regimen aimed at helping them achieve better outcomes.”
In addition to surgery, other treatment options for NETs include hormone therapy, radiation, and other interventions. The Netspot Ga-68 DOTATATE PET/CT is available at the Rocky Mountain Cancer Centers Midtown location, centrally located near downtown Denver.
About Rocky Mountain Cancer Centers
Rocky Mountain Cancer Centers is Colorado’s largest and most comprehensive provider of cancer care with 54 physicians treating patients in 19 community-based locations across the state. We are united in healing with The US Oncology Network, one of the nation’s largest networks of integrated community-based oncology practices dedicated to advancing high-quality, evidence-based patient care.
About The Neuroendocrine Institute
The Neuroendocrine Institute at Rocky Mountain Cancer Centers specializes in treating all types and stages of neuroendocrine tumors. Led by Co-Directors Dr. Allen Cohn and Dr. Eric Liu, the multidisciplinary team includes medical oncologists, surgeons, radiation oncologists, pathologists, interventional radiologists, advanced practice providers, and oncology nurses. The latest clinical trials are also available.
Media Contact
Maurine Taylor
Rocky Mountain Cancer Centers
303-930-7834
Maurine.taylor@usoncology.com